Control of transdermal permeation of hydrocortisone acetate from hydrophilic and lipophilic formulations
- PMID: 18563577
- PMCID: PMC2977040
- DOI: 10.1208/s12249-008-9107-z
Control of transdermal permeation of hydrocortisone acetate from hydrophilic and lipophilic formulations
Abstract
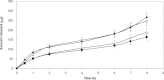
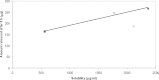
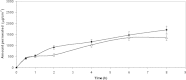
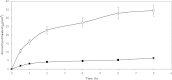
The purpose of this research was the preparation of four formulations containing hydrocortisone acetate (HCA) for topical application, including two aqueous systems (hydrophilic microemulsion and aqueous gel) and two systems with dominant hydrophobicity (hydrophobic microemulsion and ointment). The formulations were tested for the release and permeation of HCA across an animal membrane. The release of HCA was found comparable for the four systems. The two microemulsions promote permeation across an ex-vivo membrane, examined by means of a Franz cell. Hydrophobic microemulsion guarantees the highest solubility (2,370 microg/ml) and flux (133 microg/cm(2).h) of the drug, since it contains almost 40% Transcutol, a permeation enhancer. Gel and ointment provide lower solubility and flux, being the values, related to the ointment, the lowest ones (562 microg/ml and 0.4 microg/cm(2).h). Experimental results allow the conclusion that gel and ointment can be suitable when it is desirable to minimize absorption of topically applied HCA as to keep the drug restricted to the diseased area and prevent side effects of the systemic presence of HCA.
Figures
Similar articles
-
Microemulsion: a novel transdermal delivery system to facilitate skin penetration of indomethacin.Pharmazie. 2012 Apr;67(4):319-23. Pharmazie. 2012. PMID: 22570938
-
Development of matrix patches for transdermal delivery of a highly lipophilic antiestrogen.Drug Dev Ind Pharm. 2003 Aug;29(7):785-93. doi: 10.1081/ddc-120021778. Drug Dev Ind Pharm. 2003. PMID: 12906336
-
Transdermal therapeutic system of isradipine: effect of hydrophilic and hydrophobic matrix on in vitro and ex vivo characteristics.Arch Pharm Res. 2010 Jul;33(7):1025-33. doi: 10.1007/s12272-010-0708-1. Epub 2010 Jul 27. Arch Pharm Res. 2010. PMID: 20661712
-
Biomedical applications of microemulsion through dermal and transdermal route.Biomed Pharmacother. 2018 Dec;108:1477-1494. doi: 10.1016/j.biopha.2018.10.021. Epub 2018 Oct 8. Biomed Pharmacother. 2018. PMID: 30372850 Review.
-
Drug delivery systems. 6. Transdermal drug delivery.J Clin Pharmacol. 1991 May;31(5):401-18. doi: 10.1002/j.1552-4604.1991.tb01895.x. J Clin Pharmacol. 1991. PMID: 2050824 Review.
Cited by
-
Suppression of cytokine gene expression and improved therapeutic efficacy of microemulsion-based tacrolimus cream for atopic dermatitis.Drug Deliv Transl Res. 2012 Apr;2(2):129-41. doi: 10.1007/s13346-012-0060-8. Drug Deliv Transl Res. 2012. PMID: 25786721
-
Microemulsions containing medium-chain glycerides as transdermal delivery systems for hydrophilic and hydrophobic drugs.AAPS PharmSciTech. 2009;10(2):589-96. doi: 10.1208/s12249-009-9251-0. Epub 2009 May 14. AAPS PharmSciTech. 2009. PMID: 19440842 Free PMC article.
-
AI-Driven Innovation in Skin Kinetics for Transdermal Drug Delivery: Overcoming Barriers and Enhancing Precision.Pharmaceutics. 2025 Feb 2;17(2):188. doi: 10.3390/pharmaceutics17020188. Pharmaceutics. 2025. PMID: 40006555 Free PMC article. Review.
-
Fabrication and characterization of hydrocortisone loaded Dextran-Poly Lactic-co-Glycolic acid micelle.Heliyon. 2020 May 18;6(5):e03975. doi: 10.1016/j.heliyon.2020.e03975. eCollection 2020 May. Heliyon. 2020. PMID: 32455174 Free PMC article.
-
Progressing Towards the Sustainable Development of Cream Formulations.Pharmaceutics. 2020 Jul 9;12(7):647. doi: 10.3390/pharmaceutics12070647. Pharmaceutics. 2020. PMID: 32659962 Free PMC article.
References
-
- Davis A. F., Gyurik R. J., Hadgraft J., Pellet M. A., Walters K. A. Formulation strategies for modulating skin permeation. In: Walter K. A., editor. Dermatological and transdermal formulations. New York: Dekker; 2002. pp. 271–317.
-
- Orienti I., Zecchi V., Ceschel G. C., Fini A. Controlled release of hydrocortisone acetate from dermal bases. Eur. J. Drug Metab. Pharmacokinet. 1991;3:466–472. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

