Replication of clinical hepatitis B virus isolate and its application for selecting antiviral agents for chronic hepatitis B patients
- PMID: 18567076
- PMCID: PMC2716610
- DOI: 10.3748/wjg.14.3490
Replication of clinical hepatitis B virus isolate and its application for selecting antiviral agents for chronic hepatitis B patients
Abstract
Aim: To establish a cell model harboring replicative clinical hepatitis B virus (HBV) isolates and evaluate its application in individualized selection of anti-HBV agents for chronic hepatitis B (CHB) patients.
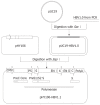
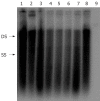
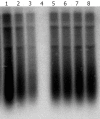
Methods: The full-length HBV genomic DNA from 8 CHB patients was amplified by polymerase chain reaction (PCR). All the patients were treated with lamivudine for at least seven months and finally became resistant to lamivudine. The amplified HBV DNA fragments were inserted into pHY106 vectors by Sap I digestion. The recombinant plasmids containing 1.1 copies of HBV genome were transiently transfected into Huh7 cell line, and the levels of HBsAg, HBeAg and intercellular HBV replicative intermediates were determined by ELISA and Southern blot analysis, respectively, with or without lamivudine and adefovir treatment. The antiviral treatment with adefovir was administered to the patients and analyzed in parallel.
Results: A total of 25 independent HBV isolates were obtained from the sera of 8 patients, each patient had at least two isolates. One isolate from each individual was selected and subcloned into pHY106 vector, including 5 isolates with YVDD mutation and 3 isolates with YIDD mutation. All recombinant plasmids harboring HBV isolates were transfected into Huh7 cells. The results indicated that HBV genome carried in HBV replicons of clinical HBV isolates could effectively replicate and express in Huh7 cells. Adefovir, but not lamivudine, inhibited HBV replication both in vitro and in vivo, and in vitro inhibition was dose-dependent.
Conclusion: The novel method described herein enables individualized selection of anti-HBV agents in clinic and is useful in future studies of antiviral therapy for CHB.
Figures


Similar articles
-
[Significance of novel HBV expression vectors in selecting antiviral drugs in clinical therapy].Zhonghua Gan Zang Bing Za Zhi. 2007 Jan;15(1):8-12. Zhonghua Gan Zang Bing Za Zhi. 2007. PMID: 17244450 Chinese.
-
In vitro susceptibility of lamivudine-resistant hepatitis B virus to adefovir and tenofovir.Antivir Ther. 2004 Jun;9(3):353-63. Antivir Ther. 2004. PMID: 15259898
-
Construction and expression of eukaryotic plasmids containing lamivudine-resistant or wild-type strains of Hepatitis B Virus genotype C.World J Gastroenterol. 2008 Jun 21;14(23):3733-8. doi: 10.3748/wjg.14.3733. World J Gastroenterol. 2008. PMID: 18595141 Free PMC article.
-
[Therapy for chronic hepatitis B: relationship between HBV genotype and efficacy or resistance of anti-viral therapy].Nihon Shokakibyo Gakkai Zasshi. 2007 Oct;104(10):1459-64. Nihon Shokakibyo Gakkai Zasshi. 2007. PMID: 17917393 Review. Japanese. No abstract available.
-
Adefovir dipivoxil in chronic hepatitis B infection.Expert Opin Pharmacother. 2004 Nov;5(11):2361-7. doi: 10.1517/14656566.5.11.2361. Expert Opin Pharmacother. 2004. PMID: 15500383 Review.
Cited by
-
Effects of Tenofovir Combined with Recombinant Human Interferon α-2b on Negative Conversion Rate, Liver Function, Immune Status, and Drug Safety in Patients with Chronic Hepatitis B: A Systematic Review and Meta-Analysis.Evid Based Complement Alternat Med. 2022 Jun 30;2022:1889628. doi: 10.1155/2022/1889628. eCollection 2022. Evid Based Complement Alternat Med. 2022. Retraction in: Evid Based Complement Alternat Med. 2022 Nov 29;2022:9791408. doi: 10.1155/2022/9791408. PMID: 35815265 Free PMC article. Retracted.
-
Hepatitis B virus mutation pattern rtL180M+A181C+M204V may contribute to entecavir resistance in clinical practice.Emerg Microbes Infect. 2019;8(1):354-365. doi: 10.1080/22221751.2019.1584018. Emerg Microbes Infect. 2019. PMID: 30866789 Free PMC article.
References
-
- Maddrey WC. Hepatitis B: an important public health issue. J Med Virol. 2000;61:362–366. - PubMed
-
- Lai CL, Ratziu V, Yuen MF, Poynard T. Viral hepatitis B. Lancet. 2003;362:2089–2094. - PubMed
-
- Villeneuve JP. The natural history of chronic hepatitis B virus infection. J Clin Virol. 2005;34 Suppl 1:S139–S142. - PubMed
-
- Cougot D, Neuveut C, Buendia MA. HBV induced carcinogenesis. J Clin Virol. 2005;34 Suppl 1:S75–S78. - PubMed
-
- Farrell GC, Teoh NC. Management of chronic hepatitis B virus infection: a new era of disease control. Intern Med J. 2006;36:100–113. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

