The trypanosome transcriptome is remodelled during differentiation but displays limited responsiveness within life stages
- PMID: 18573209
- PMCID: PMC2443814
- DOI: 10.1186/1471-2164-9-298
The trypanosome transcriptome is remodelled during differentiation but displays limited responsiveness within life stages
Abstract
Background: Trypanosomatids utilise polycistronic transcription for production of the vast majority of protein-coding mRNAs, which operates in the absence of gene-specific promoters. Resolution of nascent transcripts by polyadenylation and trans-splicing, together with specific rates of mRNA turnover, serve to generate steady state transcript levels that can differ in abundance across several orders of magnitude and can be developmentally regulated. We used a targeted oligonucleotide microarray, representing the strongly developmentally-regulated T. brucei membrane trafficking system and approximately 10% of the Trypanosoma brucei genome, to investigate both between-stage, or differentiation-dependent, transcriptome changes and within-stage flexibility in response to various challenges.
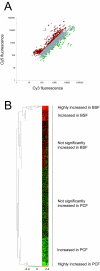
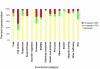
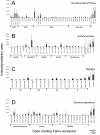
Results: 6% of the gene cohort are developmentally regulated, including several small GTPases, SNAREs, vesicle coat factors and protein kinases both consistent with and extending previous data. Therefore substantial differentiation-dependent remodeling of the trypanosome transcriptome is associated with membrane transport. Both the microarray and qRT-PCR were then used to analyse transcriptome changes resulting from specific gene over-expression, knockdown, altered culture conditions and chemical stress. Firstly, manipulation of Rab5 expression results in co-ordinate changes to clathrin protein expression levels and endocytotic activity, but no detectable changes to steady-state mRNA levels, which indicates that the effect is mediated post-transcriptionally. Secondly, knockdown of clathrin or the variant surface glycoprotein failed to perturb transcription. Thirdly, exposure to dithiothreitol or tunicamycin revealed no evidence for a classical unfolded protein response, mediated in higher eukaryotes by transcriptional changes. Finally, altered serum levels invoked little transcriptome alteration beyond changes to expression of ESAG6/7, the transferrin receptor.
Conclusion: While trypanosomes regulate mRNA abundance to effect the major changes accompanying differentiation, a given differentiated state appears transcriptionally inflexible. The implications of the absence of a transcriptome response in trypanosomes for both virulence and models of life cycle progression are discussed.
Figures



References
-
- Barry JD. Capping of variable antigen on Trypanosoma brucei, and its immunological and biological significance. J Cell Sci. 1979;37:287–302. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

