Epidermal growth factor-like domain repeat of stabilin-2 recognizes phosphatidylserine during cell corpse clearance
- PMID: 18573870
- PMCID: PMC2519725
- DOI: 10.1128/MCB.01993-07
Epidermal growth factor-like domain repeat of stabilin-2 recognizes phosphatidylserine during cell corpse clearance
Abstract
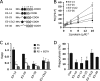
Exposure of phosphatidylserine (PS) on the cell surface occurs early during apoptosis and serves as a recognition signal for phagocytes. Clearance of apoptotic cells by a membrane PS receptor is one of the critical anti-inflammatory functions of macrophages. However, the PS binding receptors and their recognition mechanisms have not been fully investigated. Recently, we reported that stabilin-2 is a PS receptor that mediates the clearance of apoptotic cells, thus releasing the anti-inflammatory cytokine, transforming growth factor beta. In this study, we showed that epidermal growth factor (EGF)-like domain repeats (EGFrp) in stabilin-2 can directly and specifically recognize PS. The EGFrps also competitively impaired apoptotic cell uptake by macrophages in in vivo models. We also showed that calcium ions are required for stabilin-2 to mediate phagocytosis via EGFrp. Interestingly, at least four tandem repeats of EGF-like domains were required to recognize PS, and the second atypical EGF-like domain in EGFrp was critical for calcium-dependent PS recognition. Considering that PS itself is an important target molecule for both apoptotic cells and nonapoptotic cells during various cellular processes, our results should help elucidate the molecular mechanism by which apoptotic cell clearance in the human body occurs and also have implications for targeting PS externalization of nonapoptotic cells.
Figures




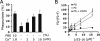


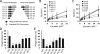
Similar articles
-
Stabilin Receptors: Role as Phosphatidylserine Receptors.Biomolecules. 2019 Aug 20;9(8):387. doi: 10.3390/biom9080387. Biomolecules. 2019. PMID: 31434355 Free PMC article. Review.
-
Stabilin-1 mediates phosphatidylserine-dependent clearance of cell corpses in alternatively activated macrophages.J Cell Sci. 2009 Sep 15;122(Pt 18):3365-73. doi: 10.1242/jcs.049569. J Cell Sci. 2009. PMID: 19726632
-
The conserved histidine in epidermal growth factor-like domains of stabilin-2 modulates pH-dependent recognition of phosphatidylserine in apoptotic cells.Int J Biochem Cell Biol. 2010 Jul;42(7):1154-63. doi: 10.1016/j.biocel.2010.03.024. Epub 2010 Apr 9. Int J Biochem Cell Biol. 2010. PMID: 20382256
-
Rapid cell corpse clearance by stabilin-2, a membrane phosphatidylserine receptor.Cell Death Differ. 2008 Jan;15(1):192-201. doi: 10.1038/sj.cdd.4402242. Epub 2007 Oct 26. Cell Death Differ. 2008. PMID: 17962816
-
The role of phosphatidylserine recognition receptors in multiple biological functions.Cell Mol Biol Lett. 2020 Mar 26;25:23. doi: 10.1186/s11658-020-00214-z. eCollection 2020. Cell Mol Biol Lett. 2020. PMID: 32226456 Free PMC article. Review.
Cited by
-
The role of nucleotides and purinergic signaling in apoptotic cell clearance - implications for chronic inflammatory diseases.Front Immunol. 2014 Dec 23;5:656. doi: 10.3389/fimmu.2014.00656. eCollection 2014. Front Immunol. 2014. PMID: 25566266 Free PMC article. Review.
-
HARE-Mediated Endocytosis of Hyaluronan and Heparin Is Targeted by Different Subsets of Three Endocytic Motifs.Int J Cell Biol. 2015;2015:524707. doi: 10.1155/2015/524707. Epub 2015 Mar 25. Int J Cell Biol. 2015. PMID: 25883656 Free PMC article.
-
Primary phagocytosis of neurons by inflamed microglia: potential roles in neurodegeneration.Front Pharmacol. 2012 Feb 28;3:27. doi: 10.3389/fphar.2012.00027. eCollection 2012. Front Pharmacol. 2012. PMID: 22403545 Free PMC article.
-
Stabilin Receptors: Role as Phosphatidylserine Receptors.Biomolecules. 2019 Aug 20;9(8):387. doi: 10.3390/biom9080387. Biomolecules. 2019. PMID: 31434355 Free PMC article. Review.
-
Molecular Chaperone Receptors: An Update.Methods Mol Biol. 2023;2693:193-208. doi: 10.1007/978-1-0716-3342-7_15. Methods Mol Biol. 2023. PMID: 37540436
References
-
- Appella, E., I. T. Weber, and F. Blasi. 1988. Structure and function of epidermal growth factor-like regions in proteins. FEBS Lett. 2311-4. - PubMed
-
- Balzar, M., I. H. Briaire-de Bruijn, H. A. Rees-Bakker, F. A. Prins, W. Helfrich, L. de Leij, G. Riethmuller, S. Alberti, S. O. Warnaar, G. J. Fleuren, and S. V. Litvinov. 2001. Epidermal growth factor-like repeats mediate lateral and reciprocal interactions of Ep-CAM molecules in homophilic adhesions. Mol. Cell. Biol. 212570-2580. - PMC - PubMed
-
- Bastiani, M. J., A. L. Harrelson, P. M. Snow, and C. S. Goodman. 1987. Expression of fasciclin I and II glycoproteins on subsets of axon pathways during neuronal development in the grasshopper. Cell 48745-755. - PubMed
-
- Bondanza, A., V. S. Zimmermann, P. Rovere-Querini, J. Turnay, I. E. Dumitriu, C. M. Stach, R. E. Voll, U. S. Gaipl, W. Bertling, E. Poschl, J. R. Kalden, A. A. Manfredi, and M. Herrmann. 2004. Inhibition of phosphatidylserine recognition heightens the immunogenicity of irradiated lymphoma cells in vivo. J. Exp. Med. 2001157-1165. - PMC - PubMed
-
- Borisenko, G. G., S. L. Iverson, S. Ahlberg, V. E. Kagan, and B. Fadeel. 2004. Milk fat globule epidermal growth factor 8 (MFG-E8) binds to oxidized phosphatidylserine: implications for macrophage clearance of apoptotic cells. Cell Death Differ. 11943-945. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous
