ATF-1 is a hypoxia-responsive transcriptional activator of skeletal muscle mitochondrial-uncoupling protein 3
- PMID: 18579531
- PMCID: PMC2517006
- DOI: 10.1074/jbc.M801236200
ATF-1 is a hypoxia-responsive transcriptional activator of skeletal muscle mitochondrial-uncoupling protein 3
Abstract
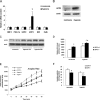
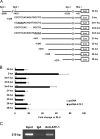
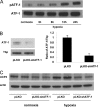
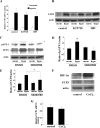
Hypoxia induces oxidative damage in skeletal muscle. Uncoupling protein 3 (UCP3) is the skeletal muscle enriched uncoupling protein and has previously been shown to confer resistance against oxidative stress. We show that hypoxia robustly up-regulates skeletal muscle UCP3 and that the absence of UCP3 in primary skeletal myocytes exacerbates hypoxia-induced reactive oxygen species generation. In this context, we reasoned that the investigation of the regulation of UCP3 may identify novel hypoxia-responsive regulatory pathways that modulate intrinsic anti-oxidant defenses. By screening a transcription factor array of 704 full-length cDNAs in murine C2C12 myoblasts following cotransfection of a murine UCP3 promoter-luciferase construct and myoD we identified numerous candidate regulatory factors that up-regulate UCP3. Active transcription factor-1 (ATF-1) was identified, and as this transcription factor is a known component of a multiprotein hypoxia-induced regulatory complex, we explored its role in hypoxia-mediated UCP3 up-regulation. Site-directed mutagenesis and chromatin immunoprecipitation assays identify a 10-bp region required for ATF-1 induction of UCP3 promoter activity. Hypoxia promotes the phosphorylation of ATF-1, and the knockdown of ATF-1 by shRNA prevents hypoxia-mediated up-regulation of UCP3. Pharmacologic inhibition of p38 MAP kinase prevents both hypoxia-mediated ATF-1 phosphorylation and UCP3 up-regulation. PKA signaling does not modulate hypoxia-induced UCP3 up-regulation and neither does HIF-1alpha activation by cobalt chloride. In conclusion, ATF-1, via p38 MAP kinase activation, functions as a novel regulatory pathway driving UCP3 expression. These data reinforce the role of ATF-1 as a hypoxia-responsive trans-activator and identifies a novel regulatory program that may modulate cellular responses to oxygen-deficit.
Figures

Similar articles
-
The preservation of in vivo phosphorylated and activated uncoupling protein 3 (UCP3) in isolated skeletal muscle mitochondria following administration of 3,4-methylenedioxymethamphetamine (MDMA aka ecstasy) to rats/mice.Mitochondrion. 2012 Jan;12(1):110-9. doi: 10.1016/j.mito.2011.03.011. Epub 2011 Mar 29. Mitochondrion. 2012. PMID: 21453795
-
SIRT1 is involved in glucocorticoid-mediated control of uncoupling protein-3 gene transcription.J Biol Chem. 2007 Nov 23;282(47):34066-76. doi: 10.1074/jbc.M707114200. Epub 2007 Sep 20. J Biol Chem. 2007. PMID: 17884810
-
Induction of endogenous uncoupling protein 3 suppresses mitochondrial oxidant emission during fatty acid-supported respiration.J Biol Chem. 2007 Oct 26;282(43):31257-66. doi: 10.1074/jbc.M706129200. Epub 2007 Aug 30. J Biol Chem. 2007. PMID: 17761668
-
Mitochondrial uncoupling protein 3 and its role in cardiac- and skeletal muscle metabolism.Physiol Behav. 2008 May 23;94(2):259-69. doi: 10.1016/j.physbeh.2007.11.039. Epub 2007 Nov 28. Physiol Behav. 2008. PMID: 18191161 Review.
-
The energetic implications of uncoupling protein-3 in skeletal muscle.Appl Physiol Nutr Metab. 2007 Oct;32(5):884-94. doi: 10.1139/H07-063. Appl Physiol Nutr Metab. 2007. PMID: 18059613 Review.
Cited by
-
HIF-1-driven skeletal muscle adaptations to chronic hypoxia: molecular insights into muscle physiology.Cell Mol Life Sci. 2015 Dec;72(24):4681-96. doi: 10.1007/s00018-015-2025-9. Epub 2015 Aug 23. Cell Mol Life Sci. 2015. PMID: 26298291 Free PMC article. Review.
-
Fatty Acids Prevent Hypoxia-Inducible Factor-1α Signaling Through Decreased Succinate in Diabetes.JACC Basic Transl Sci. 2018 Aug 28;3(4):485-498. doi: 10.1016/j.jacbts.2018.04.005. eCollection 2018 Aug. JACC Basic Transl Sci. 2018. PMID: 30175272 Free PMC article.
-
Transient upregulation of PGC-1alpha diminishes cardiac ischemia tolerance via upregulation of ANT1.J Mol Cell Cardiol. 2010 Oct;49(4):693-8. doi: 10.1016/j.yjmcc.2010.06.008. Epub 2010 Jul 1. J Mol Cell Cardiol. 2010. PMID: 20600099 Free PMC article.
-
Mild Endurance Exercise during Fasting Increases Gastrocnemius Muscle and Prefrontal Cortex Thyroid Hormone Levels through Differential BHB and BCAA-Mediated BDNF-mTOR Signaling in Rats.Nutrients. 2022 Mar 10;14(6):1166. doi: 10.3390/nu14061166. Nutrients. 2022. PMID: 35334826 Free PMC article.
-
Ionizing radiation, ion transports, and radioresistance of cancer cells.Front Physiol. 2013 Aug 14;4:212. doi: 10.3389/fphys.2013.00212. eCollection 2013. Front Physiol. 2013. PMID: 23966948 Free PMC article.
References
-
- Vidal-Puig, A. J., Grujic, D., Zhang, C. Y., Hagen, T., Boss, O., Ido, Y., Szczepanik, A., Wade, J., Mootha, V., Cortright, R., Muoio, D. M., and Lowell, B. B. (2000) J. Biol. Chem. 275 16258-16266 - PubMed
-
- Brand, M. D., and Esteves, T. C. (2005) Cell Metab. 2 85-93 - PubMed
-
- Sack, M. N. (2006) Cardiovasc Res. 72 210-219 - PubMed
-
- Garcia-Martinez, C., Sibille, B., Solanes, G., Darimont, C., Mace, K., Villarroya, F., and Gomez-Foix, A. M. (2001) Faseb. J. 15 2033-2035 - PubMed
-
- Talbot, D. A., and Brand, M. D. (2005) Biochim. Biophys. Acta 1709 150-156 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

