The polybasic region of Rho GTPases defines the cleavage by Yersinia enterocolitica outer protein T (YopT)
- PMID: 18583524
- PMCID: PMC2492827
- DOI: 10.1110/ps.035386.108
The polybasic region of Rho GTPases defines the cleavage by Yersinia enterocolitica outer protein T (YopT)
Abstract
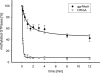
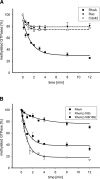
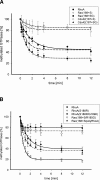
Pathogenic Yersinia strains evade the innate immune responses of the host by producing effector proteins ( Yersinia outer proteins [Yops]), which are directly injected into mammalian cells by a type III secretion system (TTSS). One of these effector proteins (YopT) disrupts the actin cytoskeleton of the host cell resulting in cell rounding. YopT is a cysteine protease that cleaves Rho proteins directly upstream of the post-translationally modified cysteine. Thereby, it releases the GTPases from the membrane leading to inactivation. Small GTPases are modified by isoprenylation of the cysteine of the CAAX box, cleavage of the -AAX tripeptide, and methylation of the cysteine. We have shown that isoprenylation and the endoproteolytic cleavage of the tripeptide of Rho GTPases are essential for YopT-induced cleavage, whereas carboxyl methylation is not required. In the present study, we post-translationally modified RhoA, Rac, Cdc42, and several mutants in vitro and characterized the YopT-induced cleavage with recombinant YopT. We show that farnesylated RhoA is a preferred substrate of YopT compared with the geranylgeranylated GTPase. Geranylgeranylated RhoA, however, is the preferred substrate for YopT-catalyzed cleavage with a threefold faster turnover rate over Rac and Cdc42. Moreover, our data indicate that the composition of the polybasic region of the GTPases defines the specificity and efficiency of the YopT-induced cleavage, and that a space between the polybasic stretch of amino acids at the C terminus and the CAAX box enhances the turnover rate of YopT-catalyzed cleavage.
Figures
Similar articles
-
Endoproteolytic processing of RhoA by Rce1 is required for the cleavage of RhoA by Yersinia enterocolitica outer protein T.Infect Immun. 2006 Mar;74(3):1712-7. doi: 10.1128/IAI.74.3.1712-1717.2006. Infect Immun. 2006. PMID: 16495543 Free PMC article.
-
Biochemical characterization of the Yersinia YopT protease: cleavage site and recognition elements in Rho GTPases.Proc Natl Acad Sci U S A. 2003 Feb 4;100(3):904-9. doi: 10.1073/pnas.252770599. Epub 2003 Jan 21. Proc Natl Acad Sci U S A. 2003. PMID: 12538863 Free PMC article.
-
Yersinia enterocolitica outer protein T (YopT).Eur J Cell Biol. 2011 Nov;90(11):955-8. doi: 10.1016/j.ejcb.2010.12.005. Epub 2011 Jan 20. Eur J Cell Biol. 2011. PMID: 21255864 Review.
-
Characterization of YopT effects on Rho GTPases in Yersinia enterocolitica-infected cells.J Biol Chem. 2003 Aug 29;278(35):33217-23. doi: 10.1074/jbc.M303349200. Epub 2003 Jun 5. J Biol Chem. 2003. PMID: 12791693
-
Modulation of Rho GTPases and the actin cytoskeleton by YopT of Yersinia.Curr Top Microbiol Immunol. 2005;291:167-75. doi: 10.1007/3-540-27511-8_9. Curr Top Microbiol Immunol. 2005. PMID: 15981463 Review.
Cited by
-
Fido, a novel AMPylation domain common to fic, doc, and AvrB.PLoS One. 2009 Jun 5;4(6):e5818. doi: 10.1371/journal.pone.0005818. PLoS One. 2009. PMID: 19503829 Free PMC article.
-
Yersinia type III effectors perturb host innate immune responses.World J Biol Chem. 2016 Feb 26;7(1):1-13. doi: 10.4331/wjbc.v7.i1.1. World J Biol Chem. 2016. PMID: 26981193 Free PMC article. Review.
-
Host manipulation by bacterial type III and type IV secretion system effector proteases.Cell Microbiol. 2021 Nov;23(11):e13384. doi: 10.1111/cmi.13384. Epub 2021 Aug 30. Cell Microbiol. 2021. PMID: 34392594 Free PMC article. Review.
-
The Pseudomonas aeruginosa T6SS Delivers a Periplasmic Toxin that Disrupts Bacterial Cell Morphology.Cell Rep. 2019 Oct 1;29(1):187-201.e7. doi: 10.1016/j.celrep.2019.08.094. Cell Rep. 2019. PMID: 31577948 Free PMC article.
-
The small GTPase RhoH is an atypical regulator of haematopoietic cells.Cell Commun Signal. 2008 Sep 29;6:6. doi: 10.1186/1478-811X-6-6. Cell Commun Signal. 2008. PMID: 18823547 Free PMC article.
References
-
- Andor, A., Essler, M., Roggenkamp, A., Heesemann, J., Aepfelbacher, M. YopE of Yersinia, a GAP for Rho-GTPases, selectively modulates Rac-dependent actin structures in endothelial cells. Cell. Microbiol. 2001;3:301–310. - PubMed
-
- Black, D.S., Marie-Cardine, A., Schraven, B., Bliska, J.B. The Yersinia tyrosine phosphatase YopH targets a novel adhesion-regulated signaling complex in macrophages. Cell. Microbiol. 2000;2:401–414. - PubMed
-
- Cornelis, G.R. The Yersinia Ysc-Yop “type III” weaponry. Nat. Rev. Mol. Cell Biol. 2002;3:742–752. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

