The chemokine CXCL12 promotes survival of postmitotic neurons by regulating Rb protein
- PMID: 18583990
- PMCID: PMC2676689
- DOI: 10.1038/cdd.2008.95
The chemokine CXCL12 promotes survival of postmitotic neurons by regulating Rb protein
Abstract
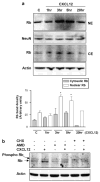
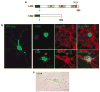
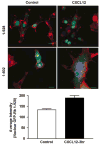
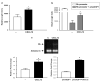
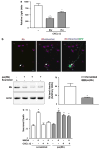
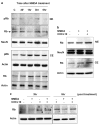
Postmitotic neurons need to keep their cell cycle under control to survive and maintain a differentiated state. This study aims to test the hypothesis that the chemokine CXCL12 regulates neuronal survival and differentiation by promoting Rb function, as suggested by previous studies showing that CXCL12 protects neurons from apoptosis induced by Rb loss. To this end, the effect of CXCL12 on Rb expression and transcriptional activity and the role of Rb in CXCL12-induced neuronal survival were studied. CXCL12 increases Rb protein and RNA levels in rat cortical neurons. The chemokine also stimulates an exogenous Rb promoter expressed in these neurons and counteracts the inhibition of the Rb promoter induced by E2F1 overexpression. Furthermore CXCL12 stimulates Rb activity as a transcription repressor. The effects of CXCL12 are mediated by its specific receptor CXCR4, and do not require the presence of glia. Finally, shRNA studies show that Rb expression is crucial to the neuroprotective activity of CXCL12 as indicated by NMDA-neurotoxicity assays. These findings suggest that proper CXCR4 stimulation in the mature CNS can prevent impairment of the Rb-E2F pathway and support neuronal survival. This is important to maintain CNS integrity in physiological conditions and prevent neuronal injury and loss typical of many neurodegenerative and neuroinflammatory conditions.
Figures
Similar articles
-
CXCL12-mediated regulation of ANP32A/Lanp, a component of the inhibitor of histone acetyl transferase (INHAT) complex, in cortical neurons.J Neuroimmune Pharmacol. 2011 Mar;6(1):163-70. doi: 10.1007/s11481-010-9228-5. Epub 2010 Jul 9. J Neuroimmune Pharmacol. 2011. PMID: 20617464 Free PMC article.
-
The chemokine receptor CXCR4 regulates cell-cycle proteins in neurons.J Neurovirol. 2003 Jun;9(3):300-14. doi: 10.1080/13550280390201010. J Neurovirol. 2003. PMID: 12775414 Free PMC article.
-
CXCL12 inhibits expression of the NMDA receptor's NR2B subunit through a histone deacetylase-dependent pathway contributing to neuronal survival.Cell Death Dis. 2010 Apr 1;1(4):e33. doi: 10.1038/cddis.2010.10. Cell Death Dis. 2010. PMID: 21364640 Free PMC article.
-
Cell cycle regulators in neural stem cells and postmitotic neurons.Neurosci Res. 2000 May;37(1):1-14. doi: 10.1016/s0168-0102(00)00101-2. Neurosci Res. 2000. PMID: 10802339 Review.
-
The CXCL12 (SDF-1)/CXCR4 chemokine axis: Oncogenic properties, molecular targeting, and synthetic and natural product CXCR4 inhibitors for cancer therapy.Chin J Nat Med. 2018 Nov;16(11):801-810. doi: 10.1016/S1875-5364(18)30122-5. Chin J Nat Med. 2018. PMID: 30502762 Review.
Cited by
-
Distinct modifications in Kv2.1 channel via chemokine receptor CXCR4 regulate neuronal survival-death dynamics.J Neurosci. 2012 Dec 5;32(49):17725-39. doi: 10.1523/JNEUROSCI.3029-12.2012. J Neurosci. 2012. PMID: 23223293 Free PMC article.
-
In vitro and in vivo experimental studies on trabecular meshwork degeneration induced by benzalkonium chloride (an American Ophthalmological Society thesis).Trans Am Ophthalmol Soc. 2012 Dec;110:40-63. Trans Am Ophthalmol Soc. 2012. PMID: 23818734 Free PMC article.
-
RB·E2F1 complex mediates DNA damage responses through transcriptional regulation of ZBRK1.J Biol Chem. 2010 Oct 22;285(43):33134-33143. doi: 10.1074/jbc.M110.143461. Epub 2010 Aug 16. J Biol Chem. 2010. PMID: 20713352 Free PMC article.
-
Novel Positive Allosteric Modulators of Glutamate Transport Have Neuroprotective Properties in an in Vitro Excitotoxic Model.ACS Chem Neurosci. 2019 Aug 21;10(8):3437-3453. doi: 10.1021/acschemneuro.9b00061. Epub 2019 Jul 11. ACS Chem Neurosci. 2019. PMID: 31257852 Free PMC article.
-
Site-specific hyperphosphorylation of pRb in HIV-induced neurotoxicity.Mol Cell Neurosci. 2011 Jun;47(2):154-65. doi: 10.1016/j.mcn.2011.04.001. Epub 2011 Apr 12. Mol Cell Neurosci. 2011. PMID: 21504794 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

