The major tegument structural protein VP22 targets areas of dispersed nucleolin and marginalized chromatin during productive herpes simplex virus 1 infection
- PMID: 18584907
- PMCID: PMC2496966
- DOI: 10.1016/j.virusres.2008.05.010
The major tegument structural protein VP22 targets areas of dispersed nucleolin and marginalized chromatin during productive herpes simplex virus 1 infection
Abstract
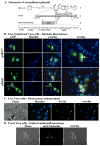
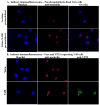
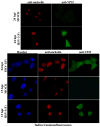
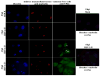
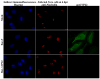
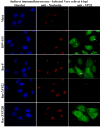
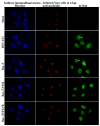
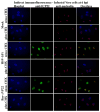
The herpes simplex virus (HSV) major tegument structural protein VP22 resides in multiple subcellular regions during productive infection. During an analysis of the molecular determinants of these localizations, we observed that a transfected fusion of the C-terminal portion of VP22, containing its pat4 nuclear localization signal, with GFP lacked nucleolar sparing compared to GFP alone. Thus, the initial goal was to determine whether VP22 associates with nucleoli. Using an optimized indirect immunofluorescence system to visualize nucleolin and viral proteins, we observed that VP22 present in VP22-expressing Vero (V49) cells "surrounded" nucleolin. These two initial findings implied that VP22 might associate directly with nucleoli. We next analyzed HSV-infected cells and observed that at late times, anti-nucleolin immune reactivity was dispersed throughout the nuclei while it retained uniform, circular staining in mock-infected cells. Time course infection experiments indicated that nucleolin initiated its transition from uniform to dispersed structures between 2 and 4 hpi. Comparison of Hoechst stained nuclei showed bright anti-nucleolin staining localized to regions of marginalized chromatin. These effects required de novo infected cell protein synthesis. A portion of VP22 detected in nuclei at 4 and 6 hpi localized to these areas of altered nucleolin and marginalized chromatin. VP22 was excluded from viral replication compartments containing the viral regulatory protein ICP22. Finally, altered nucleolin and marginalized chromatin were detected with a VP22-null virus, indicating that VP22 was not responsible for these nuclear architecture alterations. Thus, we conclude that nuclear VP22 targets unique subnuclear structures early (<6hpi) during herpes simplex virus 1 (HSV-1) infection.
Figures

Similar articles
-
Microtubule reorganization during herpes simplex virus type 1 infection facilitates the nuclear localization of VP22, a major virion tegument protein.J Virol. 2001 Sep;75(18):8697-711. doi: 10.1128/jvi.75.18.8697-8711.2001. J Virol. 2001. PMID: 11507215 Free PMC article.
-
A conserved carboxy-terminal domain in the major tegument structural protein VP22 facilitates virion packaging of a chimeric protein during productive herpes simplex virus 1 infection.Virology. 2009 May 10;387(2):449-58. doi: 10.1016/j.virol.2009.02.040. Virology. 2009. PMID: 19307008 Free PMC article.
-
Modified VP22 localizes to the cell nucleus during synchronized herpes simplex virus type 1 infection.J Virol. 1999 Aug;73(8):6769-81. doi: 10.1128/JVI.73.8.6769-6781.1999. J Virol. 1999. PMID: 10400775 Free PMC article.
-
Assembly of infectious Herpes simplex virus type 1 virions in the absence of full-length VP22.J Virol. 2000 Nov;74(21):10041-54. doi: 10.1128/jvi.74.21.10041-10054.2000. J Virol. 2000. PMID: 11024133 Free PMC article.
-
Association of the herpes simplex virus major tegument structural protein VP22 with chromatin.Biochim Biophys Acta. 2010 Mar-Apr;1799(3-4):200-6. doi: 10.1016/j.bbagrm.2009.08.002. Epub 2009 Aug 12. Biochim Biophys Acta. 2010. PMID: 19682615 Review.
Cited by
-
Enhancement of adeno-associated virus infection by mobilizing capsids into and out of the nucleolus.J Virol. 2009 Mar;83(6):2632-44. doi: 10.1128/JVI.02309-08. Epub 2008 Dec 24. J Virol. 2009. PMID: 19109385 Free PMC article.
-
Structural host-virus interactome profiling of intact infected cells.Nat Commun. 2025 Jul 21;16(1):6713. doi: 10.1038/s41467-025-61618-z. Nat Commun. 2025. PMID: 40691152 Free PMC article.
-
ORF9p phosphorylation by ORF47p is crucial for the formation and egress of varicella-zoster virus viral particles.J Virol. 2013 Mar;87(5):2868-81. doi: 10.1128/JVI.02757-12. Epub 2012 Dec 26. J Virol. 2013. PMID: 23269791 Free PMC article.
-
Alphaherpesvirus Major Tegument Protein VP22: Its Precise Function in the Viral Life Cycle.Front Microbiol. 2020 Aug 7;11:1908. doi: 10.3389/fmicb.2020.01908. eCollection 2020. Front Microbiol. 2020. PMID: 32849477 Free PMC article. Review.
-
Nucleolin is required for efficient nuclear egress of herpes simplex virus type 1 nucleocapsids.J Virol. 2010 Feb;84(4):2110-21. doi: 10.1128/JVI.02007-09. Epub 2009 Dec 2. J Virol. 2010. PMID: 19955312 Free PMC article.
References
-
- Blouin A, Blaho JA. Assessment of the subcellular localization of the herpes simplex type 1 virion structural protein VP22 in the absence of other viral gene products. 2001 submitted. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

