Characteristics and outcome of pediatric patients enrolled in phase I oncology trials
- PMID: 18586923
- PMCID: PMC6953426
- DOI: 10.1634/theoncologist.2008-0046
Characteristics and outcome of pediatric patients enrolled in phase I oncology trials
Abstract
Purpose: To describe the characteristics of pediatric subjects who enroll in phase I trials, to determine the associations between pre-enrollment characteristics and the risk for toxicity, and to analyze response and survival outcomes.
Experimental design: Pre-enrollment characteristics and study outcomes were retrospectively analyzed for children with refractory solid tumors treated in one of 16 phase I trials with similar eligibility criteria at the National Cancer Institute between 1992 and 2005.
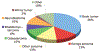
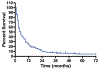
Results: The 262 subjects analyzed had received a median of two (range, 0-9) prior chemotherapy regimens, and were on one (range, 0-12) concomitant medication. The Eastern Cooperative Oncology Group performance status scores for subjects were 0 (29%), 1 (48%), and 2 (19%); 19% had received a prior stem cell transplantation and 73% had received prior radiation. Approximately 90% of subjects were evaluable for the primary trial endpoints (toxicity and pharmacokinetics). Seventeen percent of subjects experienced a dose-limiting toxicity (DLT), 5% discontinued the study drug because of toxicity, and a drug-related death occurred in one subject (0.4%). Variables associated with a higher risk for developing a DLT, by multiple logistic regression analysis, were drug dose and prior radiation, for myelosuppressive agents, and drug dose and performance status, for nonmyelosuppressive agents. The complete and partial response rate was 4%; however, 17% of subjects had stable disease (received three or more cycles). The median overall survival time from the time of enrollment was five months.
Conclusions: Primary trial objectives are achieved in approximately 90% of subjects with the standard phase I trial design and eligibility criteria despite the intensification of frontline and salvage therapies in pediatric subjects with cancer.
Conflict of interest statement
Figures
Similar articles
-
Historical time to disease progression and progression-free survival in patients with recurrent/refractory neuroblastoma treated in the modern era on Children's Oncology Group early-phase trials.Cancer. 2017 Dec 15;123(24):4914-4923. doi: 10.1002/cncr.30934. Epub 2017 Sep 8. Cancer. 2017. PMID: 28885700 Free PMC article. Review.
-
Regional deep hyperthermia for salvage treatment of children and adolescents with refractory or recurrent non-testicular malignant germ-cell tumours: an open-label, non-randomised, single-institution, phase 2 study.Lancet Oncol. 2013 Aug;14(9):843-52. doi: 10.1016/S1470-2045(13)70271-7. Epub 2013 Jul 1. Lancet Oncol. 2013. PMID: 23823158 Clinical Trial.
-
Predictors of response, progression-free survival, and overall survival using NANT Response Criteria (v1.0) in relapsed and refractory high-risk neuroblastoma.Pediatr Blood Cancer. 2018 May;65(5):e26940. doi: 10.1002/pbc.26940. Epub 2018 Jan 19. Pediatr Blood Cancer. 2018. PMID: 29350464 Free PMC article.
-
A Systematic Review of Pediatric Phase I Trials in Oncology: Toxicity and Outcomes in the Era of Targeted Therapies.Oncologist. 2020 Jun;25(6):532-540. doi: 10.1634/theoncologist.2019-0615. Epub 2020 Jan 14. Oncologist. 2020. PMID: 31943534 Free PMC article.
-
Ifosfamide and vinorelbine is an effective reinduction regimen in children with refractory/relapsed Hodgkin lymphoma, AHOD00P1: a children's oncology group report.Pediatr Blood Cancer. 2015 Jan;62(1):60-4. doi: 10.1002/pbc.25205. Epub 2014 Oct 12. Pediatr Blood Cancer. 2015. PMID: 25308760 Free PMC article. Clinical Trial.
Cited by
-
Clinical characteristics and outcomes of pediatric oncology patients with aggressive biology enrolled in phase I clinical trials designed for adults: the university of Texas MD anderson cancer center experience.Oncoscience. 2014 Jul 27;1(7):522-530. doi: 10.18632/oncoscience.68. Oncoscience. 2014. PMID: 25587555 Free PMC article.
-
Medulloblastoma in children and adolescents: a systematic review of contemporary phase I and II clinical trials and biology update.Cancer Med. 2017 Nov;6(11):2606-2624. doi: 10.1002/cam4.1171. Epub 2017 Oct 4. Cancer Med. 2017. PMID: 28980418 Free PMC article.
-
Physician and nurse beliefs of phase 1 trials in pediatric oncology.Cancer Nurs. 2014 Sep-Oct;37(5):E48-52. doi: 10.1097/NCC.0000000000000099. Cancer Nurs. 2014. PMID: 25140639 Free PMC article.
-
Opportunities and Challenges in Drug Development for Pediatric Cancers.Cancer Discov. 2021 Mar;11(3):545-559. doi: 10.1158/2159-8290.CD-20-0779. Epub 2020 Dec 4. Cancer Discov. 2021. PMID: 33277309 Free PMC article. Review.
-
Early phase study enrollment in Canadian children with cancer near end of life: A retrospective cohort study from Cancer in Young People in Canada.Cancer. 2025 Jun 15;131(12):e35942. doi: 10.1002/cncr.35942. Cancer. 2025. PMID: 40515601 Free PMC article.
References
-
- Smith M, Bernstein M, Bleyer WA et al. Conduct of phase I trials in children with cancer. J Clin Oncol 1998;16:966–978. - PubMed
-
- Adamson PC, Blaney SM. New approaches to drug development in pediatric oncology. Cancer J 2005;11:324–330. - PubMed
-
- Balis FM. The Challenge of developing new therapies for childhood cancers. The Oncologist 1997;2:I–II. - PubMed
-
- Carlson L, Ho P, Smith M et al. Pediatric phase I drug tolerance: A review and comparison of recent adult and pediatric phase I trials. J Pediatr Hematol Oncol 1996;18:250–256. - PubMed
-
- Shah S, Weitman S, Langevin AM et al. Phase I therapy trials in children with cancer. J Pediatr Hematol Oncol 1998;20:431–438. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

