Bacillus subtilis spores germinate in the chicken gastrointestinal tract
- PMID: 18586965
- PMCID: PMC2519260
- DOI: 10.1128/AEM.00580-08
Bacillus subtilis spores germinate in the chicken gastrointestinal tract
Abstract
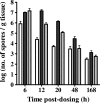
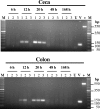
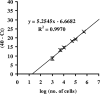
A number of poultry probiotics contain bacterial spores. In this study, orally administered spores of Bacillus subtilis germinated in the gastrointestinal (GI) tracts of chicks. Furthermore, 20 h after spores were administered, vegetative cells outnumbered spores throughout the GI tract. This demonstrates that spore-based probiotics may function in this host through metabolically active mechanisms.
Figures

Similar articles
-
Germination and outgrowth of Bacillus subtilis and Bacillus licheniformis spores in the gastrointestinal tract of pigs.J Appl Microbiol. 2008 Apr;104(4):1025-33. doi: 10.1111/j.1365-2672.2007.03633.x. Epub 2007 Nov 15. J Appl Microbiol. 2008. PMID: 18005348
-
Evaluation of germination, distribution, and persistence of Bacillus subtilis spores through the gastrointestinal tract of chickens.Poult Sci. 2014 Jul;93(7):1793-800. doi: 10.3382/ps.2013-03809. Epub 2014 May 7. Poult Sci. 2014. PMID: 24812242
-
Isolation and characterization of Bacillus subtilis CH16 strain from chicken gastrointestinal tracts for use as a feed supplement to promote weight gain in broilers.Lett Appl Microbiol. 2015 Jun;60(6):580-8. doi: 10.1111/lam.12411. Epub 2015 Mar 29. Lett Appl Microbiol. 2015. PMID: 25754534
-
Proteomics and microscopy tools for the study of antimicrobial resistance and germination mechanisms of bacterial spores.Food Microbiol. 2019 Aug;81:89-96. doi: 10.1016/j.fm.2018.03.006. Epub 2018 Mar 14. Food Microbiol. 2019. PMID: 30910091 Review.
-
Origin of bacterial spores contaminating foods.Food Microbiol. 2011 Apr;28(2):177-82. doi: 10.1016/j.fm.2010.07.008. Epub 2010 Jul 15. Food Microbiol. 2011. PMID: 21315971 Review.
Cited by
-
Development and Evaluation of a Commercial Direct-Fed Microbial (Zymospore®) on the Fecal Microbiome and Growth Performance of Broiler Chickens under Experimental Challenge Conditions.Animals (Basel). 2022 Jun 2;12(11):1436. doi: 10.3390/ani12111436. Animals (Basel). 2022. PMID: 35681899 Free PMC article.
-
Effect of Different Concentrations of Bacillus subtilis on Immune Response of Broiler Chickens.Probiotics Antimicrob Proteins. 2011 Mar;3(1):8-14. doi: 10.1007/s12602-011-9063-x. Probiotics Antimicrob Proteins. 2011. PMID: 26781494
-
Combined Use of Bacillus subtilis yb-114,246 and Bacillus licheniformis yb-214,245 Improves Body Growth Performance of Chinese Huainan Partridge Shank Chickens by Enhancing Intestinal Digestive Profiles.Probiotics Antimicrob Proteins. 2021 Apr;13(2):327-342. doi: 10.1007/s12602-020-09691-2. Probiotics Antimicrob Proteins. 2021. PMID: 32783087
-
Bacillus amyloliquefaciens CECT 5940 improves performance and gut function in broilers fed different levels of protein and/or under necrotic enteritis challenge.Anim Nutr. 2021 Mar;7(1):185-197. doi: 10.1016/j.aninu.2020.05.004. Epub 2020 Aug 23. Anim Nutr. 2021. PMID: 33997347 Free PMC article.
-
A synbiotic concept containing spore-forming Bacillus strains and a prebiotic fiber blend consistently enhanced metabolic activity by modulation of the gut microbiome in vitro.Int J Pharm X. 2019 Jul 6;1:100021. doi: 10.1016/j.ijpx.2019.100021. eCollection 2019 Dec. Int J Pharm X. 2019. PMID: 31517286 Free PMC article.
References
-
- Anadon, A., M. R. Martinez-Larranaga, and M. Aranzazu Martinez. 2006. Probiotics for animal nutrition in the European Union. Regulation and safety assessment. Regul. Toxicol. Pharmacol. 45:91-95. - PubMed
-
- Balu, S., and P. Kaiser. 2003. Avian interleukin-12beta (p40): cloning and characterization of the cDNA and gene. J. Interferon Cytokine Res. 23:699-707. - PubMed
-
- Bunyan, J., L. Jeffries, J. R. Sayers, A. L. Gulliver, and K. Coleman. 1977. Antimicrobial substances and chick growth promotion: the growth promoting activities of antimicrobial substances, including fifty-two used either in therapy or as dietary feed additives. Br. Poult. Sci. 18:283-294.
-
- Cartman, S. T., and R. M. La Ragione. 2004. Spore probiotics as animal feed supplements, p. 155-161. In E. Ricca, A. O. Henriques, and S. M. Cutting (ed.), Bacterial spore formers: probiotics and emerging applications. Horizon Bioscience, Norfolk, United Kingdom.
-
- Cartman, S. T., R. M. La Ragione, and M. J. Woodward. 2007. Bacterial spore formers as probiotics for poultry. Food Sci. Tech. Bull. 4:21-30.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

