Integrins alpha1beta1 and alpha2beta1 are receptors for the rotavirus enterotoxin
- PMID: 18587047
- PMCID: PMC3021436
- DOI: 10.1073/pnas.0803934105
Integrins alpha1beta1 and alpha2beta1 are receptors for the rotavirus enterotoxin
Abstract
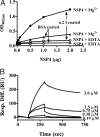
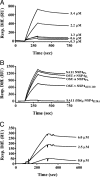
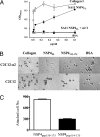
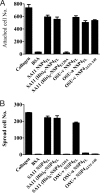
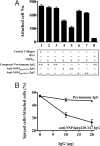
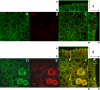
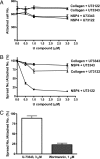
Rotavirus NSP4 is a viral enterotoxin capable of causing diarrhea in neonatal mice. This process is initiated by the binding of extracellular NSP4 to target molecule(s) on the cell surface that triggers a signaling cascade leading to diarrhea. We now report that the integrins alpha1beta1 and alpha2beta1 are receptors for NSP4. NSP4 specifically binds to the alpha1 and alpha2 I domains with apparent K(d) = 1-2.7 muM. Binding is mediated by the I domain metal ion-dependent adhesion site motif, requires Mg(2+) or Mn(2+), is abolished with EDTA, and an NSP4 point mutant, E(120)A, fails to bind alpha2 integrin I domain. NSP4 has two distinct integrin interaction domains. NSP4 amino acids 114-130 are essential for binding to the I domain, and NSP4 peptide 114-135 blocks binding of the natural ligand, collagen I, to integrin alpha2. NSP4 amino acids 131-140 are not associated with the initial binding to the I domain, but elicit signaling that leads to the spreading of attached C2C12-alpha2 cells, mouse myoblast cells stably expressing the human alpha2 integrin. NSP4 colocalizes with integrin alpha2 on the basolateral surface of rotavirus-infected polarized intestinal epithelial (Caco-2) cells as well as surrounding noninfected cells. NSP4 mutants that fail to bind or signal through integrin alpha2 were attenuated in diarrhea induction in neonatal mice. These results indicate that NSP4 interaction with integrin alpha1 and alpha2 is an important component of enterotoxin function and rotavirus pathogenesis, further distinguishing this viral virulence factor from other microbial enterotoxins.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Rotavirus enterotoxin NSP4 binds to the extracellular matrix proteins laminin-beta3 and fibronectin.J Virol. 2004 Sep;78(18):10045-53. doi: 10.1128/JVI.78.18.10045-10053.2004. J Virol. 2004. PMID: 15331737 Free PMC article.
-
Epitope mapping and use of epitope-specific antisera to characterize the VP5* binding site in rotavirus SA11 NSP4.Virology. 2008 Mar 30;373(1):211-28. doi: 10.1016/j.virol.2007.11.021. Epub 2007 Dec 31. Virology. 2008. PMID: 18164740 Free PMC article.
-
The rotavirus enterotoxin NSP4 directly interacts with the caveolar structural protein caveolin-1.J Virol. 2006 Mar;80(6):2842-54. doi: 10.1128/JVI.80.6.2842-2854.2006. J Virol. 2006. PMID: 16501093 Free PMC article.
-
How do the rotavirus NSP4 and bacterial enterotoxins lead differently to diarrhea?Virol J. 2007 Mar 21;4:31. doi: 10.1186/1743-422X-4-31. Virol J. 2007. PMID: 17376232 Free PMC article. Review.
-
Rotavirus NSP4: a multifunctional viral enterotoxin.Viral Immunol. 2005;18(1):27-40. doi: 10.1089/vim.2005.18.27. Viral Immunol. 2005. PMID: 15802952 Review. No abstract available.
Cited by
-
Equine Rotavirus A under the One Health Lens: Potential Impacts on Public Health.Viruses. 2024 Jan 16;16(1):130. doi: 10.3390/v16010130. Viruses. 2024. PMID: 38257830 Free PMC article. Review.
-
Rotaviruses: From Pathogenesis to Disease Control-A Critical Review.Viruses. 2022 Apr 22;14(5):875. doi: 10.3390/v14050875. Viruses. 2022. PMID: 35632617 Free PMC article. Review.
-
Chloride channel inhibition by a red wine extract and a synthetic small molecule prevents rotaviral secretory diarrhoea in neonatal mice.Gut. 2014 Jul;63(7):1120-9. doi: 10.1136/gutjnl-2013-305663. Epub 2013 Sep 19. Gut. 2014. PMID: 24052273 Free PMC article.
-
Pathophysiological Consequences of Calcium-Conducting Viroporins.Annu Rev Virol. 2015 Nov;2(1):473-96. doi: 10.1146/annurev-virology-100114-054846. Annu Rev Virol. 2015. PMID: 26958925 Free PMC article. Review.
-
Rotavirus infection of cells in culture induces activation of RhoA and changes in the actin and tubulin cytoskeleton.PLoS One. 2012;7(10):e47612. doi: 10.1371/journal.pone.0047612. Epub 2012 Oct 17. PLoS One. 2012. PMID: 23082182 Free PMC article.
References
-
- Jayaram H, Estes MK, Prasad BV. Emerging themes in rotavirus cell entry, genome organization, transcription and replication. Virus Res. 2004;101:67–81. - PubMed
-
- Glass RI, Parashar UD. The promise of new rotavirus vaccines. N Engl J Med. 2006;354:75–77. - PubMed
-
- Blutt SE, et al. Rotavirus antigenaemia and viraemia: A common event? Lancet. 2003;362:1445–1449. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

