Human herpesvirus-8 infection of primary pulmonary microvascular endothelial cells
- PMID: 18587055
- PMCID: PMC2586046
- DOI: 10.1165/rcmb.2007-0368OC
Human herpesvirus-8 infection of primary pulmonary microvascular endothelial cells
Abstract
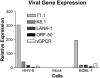
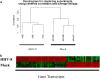
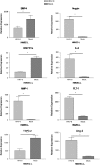
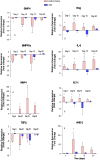
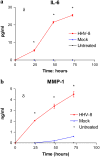
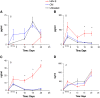
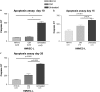
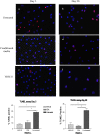
Human herpesvirus-8 (HHV-8) is the causative agent of Kaposi's sarcoma and is associated with the angioproliferative disorders primary effusion lymphoma and multicentric Castleman's disease. Evidence of HHV-8 infection within the pulmonary vasculature of patients with idiopathic pulmonary arterial hypertension (IPAH) has been described. We hypothesize that HHV-8 infection of pulmonary microvascular endothelial cells results in an apoptotic-resistant phenotype characteristic of severe pulmonary arterial hypertension. Our objective was to investigate the ability of HHV-8 to infect human pulmonary microvascular endothelial cells in vitro and characterize the phenotypic effect of this infection. Human pulmonary microvascular endothelial cells were exposed to HHV-8 using two methods (direct virus and co-culture technique). The presence of lytic and latent infection was confirmed. Changes in endothelial cell gene and protein expression and effects on cellular apoptosis were measured. HHV-8 can both lytically and latently infect primary human pulmonary microvascular endothelial cells in vitro. HHV-8 infection results in significant changes in gene expression, including alterations of pathways important to cellular apoptosis. HHV-8 infection also alters expression of genes integral to the bone morphogenic protein pathway, including down-regulation of bone morphogenic protein-4. Other genes previously implicated in the development of PAH are affected by HHV-8 infection, and cells infected with HHV-8 are resistant to apoptosis.
Figures


References
-
- Humbert M, Morrell NW, Archer SL, Stenmark KR, MacLean MR, Lang IM, Christman BW, Weir EK, Eickelberg O, Voelkel NF, et al. Cellular and molecular pathobiology of pulmonary arterial hypertension. J Am Coll Cardiol 2004;43:13S–24S. - PubMed
-
- Voelkel NF, Cool C, Lee SD, Wright L, Geraci MW, Tuder RM. Primary pulmonary hypertension between inflammation and cancer. Chest 1998;114:225S–230S. - PubMed
-
- Lane KB, Machado RD, Pauciulo MW, Thomson JR, Phillips JA III, Loyd JE, Nichols WC, Trembath RC. Heterozygous germline mutations in BMPR2, encoding a TGF-beta receptor, cause familial primary pulmonary hypertension. The International PPH Consortium. Nat Genet 2000;26:81–84. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

