RAWUL: a new ubiquitin-like domain in PRC1 ring finger proteins that unveils putative plant and worm PRC1 orthologs
- PMID: 18588675
- PMCID: PMC2447854
- DOI: 10.1186/1471-2164-9-308
RAWUL: a new ubiquitin-like domain in PRC1 ring finger proteins that unveils putative plant and worm PRC1 orthologs
Abstract
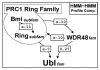
Background: Polycomb group (PcG) proteins are a set of chromatin-modifying proteins that play a key role in epigenetic gene regulation. The PcG proteins form large multiprotein complexes with different activities. The two best-characterized PcG complexes are the PcG repressive complex 1 (PRC1) and 2 (PRC2) that respectively possess histone 2A lysine 119 E3 ubiquitin ligase and histone 3 lysine 27 methyltransferase activities. While PRC2-like complexes are conserved throughout the eukaryotic kingdoms, PRC1-like complexes have only been described in Drosophila and vertebrates. Since both complexes are required for the gene silencing mechanism in Drosophila and vertebrates, how PRC1 function is realized in organisms that apparently lack PRC1 such as plants, is so far unknown. In vertebrates, PRC1 includes three proteins, Ring1B, Ring1A, and Bmi-1 that form an E3 ubiquitin ligase complex. These PRC1 proteins have an N-terminally located Ring finger domain associated to a poorly characterized conserved C-terminal region.
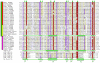
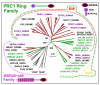
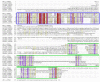
Results: We obtained statistically significant evidences of sequence similarity between the C-terminal region of the PRC1 Ring finger proteins and the ubiquitin (Ubq)-like family proteins, thus defining a new Ubq-like domain, the RAWUL domain. In addition, our analysis revealed the existence of plant and worm proteins that display the conserved combination of a Ring finger domain at the N-terminus and a RAWUL domain at the C-terminus.
Conclusion: Analysis of the conserved domain architecture among PRC1 Ring finger proteins revealed the existence of long sought PRC1 protein orthologs in these organisms, suggesting the functional conservation of PRC1 throughout higher eukaryotes.
Figures
Similar articles
-
Structure and E3-ligase activity of the Ring-Ring complex of polycomb proteins Bmi1 and Ring1b.EMBO J. 2006 Jun 7;25(11):2465-74. doi: 10.1038/sj.emboj.7601144. Epub 2006 May 18. EMBO J. 2006. PMID: 16710298 Free PMC article.
-
Evolution and conservation of polycomb repressive complex 1 core components and putative associated factors in the green lineage.BMC Genomics. 2019 Jun 28;20(1):533. doi: 10.1186/s12864-019-5905-9. BMC Genomics. 2019. PMID: 31253095 Free PMC article.
-
Interaction proteomics analysis of polycomb proteins defines distinct PRC1 complexes in mammalian cells.Mol Cell Proteomics. 2011 Apr;10(4):M110.002642. doi: 10.1074/mcp.M110.002642. Epub 2011 Jan 31. Mol Cell Proteomics. 2011. PMID: 21282530 Free PMC article.
-
Molecular architecture of polycomb repressive complexes.Biochem Soc Trans. 2017 Feb 8;45(1):193-205. doi: 10.1042/BST20160173. Biochem Soc Trans. 2017. PMID: 28202673 Free PMC article. Review.
-
Roles of Polycomb complexes in regulating gene expression and chromatin structure in plants.Plant Commun. 2021 Nov 26;3(1):100267. doi: 10.1016/j.xplc.2021.100267. eCollection 2022 Jan 10. Plant Commun. 2021. PMID: 35059633 Free PMC article. Review.
Cited by
-
Structure of the polycomb group protein PCGF1 in complex with BCOR reveals basis for binding selectivity of PCGF homologs.Structure. 2013 Apr 2;21(4):665-71. doi: 10.1016/j.str.2013.02.013. Epub 2013 Mar 21. Structure. 2013. PMID: 23523425 Free PMC article.
-
Epigenetic modifications in plants: an evolutionary perspective.Curr Opin Plant Biol. 2011 Apr;14(2):179-86. doi: 10.1016/j.pbi.2010.12.002. Epub 2011 Jan 11. Curr Opin Plant Biol. 2011. PMID: 21233005 Free PMC article. Review.
-
Arabidopsis MSI1 connects LHP1 to PRC2 complexes.EMBO J. 2013 Jul 17;32(14):2073-85. doi: 10.1038/emboj.2013.145. Epub 2013 Jun 18. EMBO J. 2013. PMID: 23778966 Free PMC article.
-
Molecular and functional characterization of broccoli EMBRYONIC FLOWER 2 genes.Plant Cell Physiol. 2012 Jul;53(7):1217-31. doi: 10.1093/pcp/pcs063. Epub 2012 Apr 25. Plant Cell Physiol. 2012. PMID: 22537758 Free PMC article.
-
Characterization of DNA methylation reader proteins in Arabidopsis thaliana.Genome Res. 2024 Dec 23;34(12):2229-2243. doi: 10.1101/gr.279379.124. Genome Res. 2024. PMID: 39632087 Free PMC article.
References
-
- Ringrose L, Paro R. Epigenetic regulation of cellular memory by the Polycomb and Trithorax group proteins. Annu Rev Genet. 2004;38:413–43. - PubMed
-
- Martinez A, Cavalli G. The role of polycomb group proteins in cell cycle regulation during development. Cell Cycle. 2006;5:1189–97. - PubMed
-
- Valk-Lingbeek M, Bruggeman S, van Lohuizen M. Stem cells and cancer; the polycomb connection. Cell. 2004;118:409–18. - PubMed
-
- Pien S, Grossniklaus U. Polycomb group and trithorax group proteins in Arabidopsis. Biochim Biophys Acta. 2007;1769:375–82. - PubMed
-
- Calonje M, Sung ZR. Complexity beneath the silence. Curr Opin Plant Biol. 2006;9:530–537. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

