Structural basis of guanine nucleotide exchange mediated by the T-cell essential Vav1
- PMID: 18589439
- PMCID: PMC2620086
- DOI: 10.1016/j.jmb.2008.05.024
Structural basis of guanine nucleotide exchange mediated by the T-cell essential Vav1
Abstract
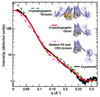
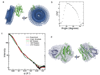
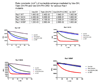
The guanine nucleotide exchange factor (GEF) Vav1 plays an important role in T-cell activation and tumorigenesis. In the GEF superfamily, Vav1 has the ability to interact with multiple families of Rho GTPases. The structure of the Vav1 DH-PH-CRD/Rac1 complex to 2.6 A resolution reveals a unique intramolecular network of contacts between the Vav1 cysteine-rich domain (CRD) and the C-terminal helix of the Vav1 Dbl homology (DH) domain. These unique interactions stabilize the Vav1 DH domain for its intimate association with the Switch II region of Rac1 that is critical for the displacement of the guanine nucleotide. Small angle x-ray scattering (SAXS) studies support this domain arrangement for the complex in solution. Further, mutational analyses confirms that the atypical CRD is critical for maintaining both optimal guanine nucleotide exchange activity and broader specificity of Vav family GEFs. Taken together, the data outline the detailed nature of Vav1's ability to contact a range of Rho GTPases using a novel protein-protein interaction network.
Figures









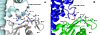
References
-
- Rossman KL, Der CJ, Sondek J. GEF means go: turning on RHO GTPases with guanine nucleotide-exchange factors. Nat Rev Mol Cell Biol. 2005;6:167–180. - PubMed
-
- Schapira V, Lazer G, Katzav S. Osteopontin is an oncogenic Vav1-but not wild-type Vav1-responsive gene: implications for fibroblast transformation. Cancer Res. 2006;66:6183–6191. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

