A Mycobacterium tuberculosis Rpf double-knockout strain exhibits profound defects in reactivation from chronic tuberculosis and innate immunity phenotypes
- PMID: 18591237
- PMCID: PMC2519441
- DOI: 10.1128/IAI.01735-07
A Mycobacterium tuberculosis Rpf double-knockout strain exhibits profound defects in reactivation from chronic tuberculosis and innate immunity phenotypes
Abstract
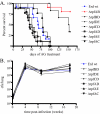
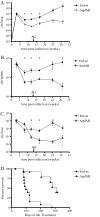
Resuscitation-promoting factors (Rpfs), apparent peptidoglycan hydrolases, have been implicated in the reactivation of dormant bacteria. We previously demonstrated that deletion of rpfB impaired reactivation of Mycobacterium tuberculosis in a mouse model. Because M. tuberculosis encodes five Rpf paralogues, redundant functions among the family members might obscure rpf single-knockout phenotypes. A series of rpf double knockouts were therefore generated. One double mutant, DeltarpfAB, exhibited several striking phenotypes. Consistent with the proposed cell wall-modifying function of Rpfs, DeltarpfAB exhibited an altered colony morphology. Although DeltarpfAB grew comparably to the parental strain in axenic culture, in vivo it exhibited deficiency in reactivation induced in C57BL/6 mice by the administration of nitric oxide synthase inhibitor (aminoguanidine) or by CD4(+) T-cell depletion. Notably, the reactivation deficiency of DeltarpfAB was more severe than that of DeltarpfB in aminoguanidine-treated mice. A similar deficiency was observed in DeltarpfAB reactivation from a drug-induced apparently sterile state in infected NOS2(-/-) mice upon cessation of antimycobacterial therapy. Secondly, DeltarpfAB showed a persistence defect not seen with the DeltarpfB or DeltarpfA single mutants. Interestingly, DeltarpfAB exhibited impaired growth in primary mouse macrophages and induced higher levels of the proinflammatory cytokines tumor necrosis factor alpha and interleukin 6. Simultaneous reintroduction of rpfA and rpfB into the double-knockout strain complemented the colony morphology and macrophage cytokine secretion phenotypes. Phenotypes related to cell wall composition and macrophage responses suggest that M. tuberculosis Rpfs may influence the outcome of reactivation, in part, by modulating innate immune responses to the bacterium.
Figures

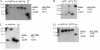



References
-
- Bardarov, S., S. Bardarov, Jr., M. S. Pavelka, Jr., V. Sambandamurthy, M. Larsen, J. Tufariello, J. Chan, G. Hatfull, and W. R. Jacobs, Jr. 2002. Specialized transduction: an efficient method for generating marked and unmarked targeted gene disruptions in Mycobacterium tuberculosis, M. bovis BCG and M. smegmatis. Microbiology 1483007-3017. - PubMed
-
- Berg, C. M., N. B. Vartak, G. Wang, X. Xu, L. Liu, D. J. MacNeil, K. M. Gewain, L. A. Wiater, and D. E. Berg. 1992. The m gamma delta-1 element, a small gamma delta (Tn1000) derivative useful for plasmid mutagenesis, allele replacement and DNA sequencing. Gene 1139-16. - PubMed
-
- Cohen-Gonsaud, M., P. Barthe, C. Bagneris, B. Henderson, J. Ward, C. Roumestand, and N. H. Keep. 2005. The structure of a resuscitation-promoting factor domain from Mycobacterium tuberculosis shows homology to lysozymes. Nat. Struct. Mol. Biol. 12270-273. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

