A close association of RyRs with highly dense clusters of Ca2+-activated Cl- channels underlies the activation of STICs by Ca2+ sparks in mouse airway smooth muscle
- PMID: 18591421
- PMCID: PMC2442178
- DOI: 10.1085/jgp.200709933
A close association of RyRs with highly dense clusters of Ca2+-activated Cl- channels underlies the activation of STICs by Ca2+ sparks in mouse airway smooth muscle
Abstract
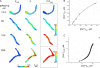
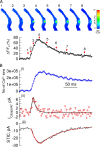
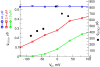
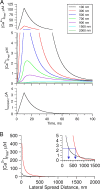
Ca(2+) sparks are highly localized, transient releases of Ca(2+) from sarcoplasmic reticulum through ryanodine receptors (RyRs). In smooth muscle, Ca(2+) sparks trigger spontaneous transient outward currents (STOCs) by opening nearby clusters of large-conductance Ca(2+)-activated K(+) channels, and also gate Ca(2+)-activated Cl(-) (Cl((Ca))) channels to induce spontaneous transient inward currents (STICs). While the molecular mechanisms underlying the activation of STOCs by Ca(2+) sparks is well understood, little information is available on how Ca(2+) sparks activate STICs. In the present study, we investigated the spatial organization of RyRs and Cl((Ca)) channels in spark sites in airway myocytes from mouse. Ca(2+) sparks and STICs were simultaneously recorded, respectively, with high-speed, widefield digital microscopy and whole-cell patch-clamp. An image-based approach was applied to measure the Ca(2+) current underlying a Ca(2+) spark (I(Ca(spark))), with an appropriate correction for endogenous fixed Ca(2+) buffer, which was characterized by flash photolysis of NPEGTA. We found that I(Ca(spark)) rises to a peak in 9 ms and decays with a single exponential with a time constant of 12 ms, suggesting that Ca(2+) sparks result from the nonsimultaneous opening and closure of multiple RyRs. The onset of the STIC lags the onset of the I(Ca(spark)) by less than 3 ms, and its rising phase matches the duration of the I(Ca(spark)). We further determined that Cl((Ca)) channels on average are exposed to a [Ca(2+)] of 2.4 microM or greater during Ca(2+) sparks. The area of the plasma membrane reaching this level is <600 nm in radius, as revealed by the spatiotemporal profile of [Ca(2+)] produced by a reaction-diffusion simulation with measured I(Ca(spark)). Finally we estimated that the number of Cl((Ca)) channels localized in Ca(2+) spark sites could account for all the Cl((Ca)) channels in the entire cell. Taken together these results lead us to propose a model in which RyRs and Cl((Ca)) channels in Ca(2+) spark sites localize near to each other, and, moreover, Cl((Ca)) channels concentrate in an area with a radius of approximately 600 nm, where their density reaches as high as 300 channels/microm(2). This model reveals that Cl((Ca)) channels are tightly controlled by Ca(2+) sparks via local Ca(2+) signaling.
Figures



Similar articles
-
Local Ca(2+) transients and distribution of BK channels and ryanodine receptors in smooth muscle cells of guinea-pig vas deferens and urinary bladder.J Physiol. 2001 Jul 15;534(Pt. 2):313-26. doi: 10.1111/j.1469-7793.2001.t01-3-00313.x. J Physiol. 2001. PMID: 11454953 Free PMC article.
-
Dynamics of signaling between Ca(2+) sparks and Ca(2+)- activated K(+) channels studied with a novel image-based method for direct intracellular measurement of ryanodine receptor Ca(2+) current.J Gen Physiol. 2000 Dec;116(6):845-64. doi: 10.1085/jgp.116.6.845. J Gen Physiol. 2000. PMID: 11099351 Free PMC article.
-
Spatial organization of RYRs and BK channels underlying the activation of STOCs by Ca(2+) sparks in airway myocytes.J Gen Physiol. 2011 Aug;138(2):195-209. doi: 10.1085/jgp.201110626. Epub 2011 Jul 11. J Gen Physiol. 2011. PMID: 21746845 Free PMC article.
-
Signaling between SR and plasmalemma in smooth muscle: sparks and the activation of Ca2+-sensitive ion channels.Cell Calcium. 2003 Sep;34(3):211-29. doi: 10.1016/s0143-4160(03)00124-6. Cell Calcium. 2003. PMID: 12887969 Review.
-
Sarcoplasmic reticulum and membrane currents.Novartis Found Symp. 2002;246:189-203; discussion 203-7, 221-7. Novartis Found Symp. 2002. PMID: 12164309 Review.
Cited by
-
Na+/Ca2 + Exchange and Pacemaker Activity of Interstitial Cells of Cajal.Front Physiol. 2020 Mar 18;11:230. doi: 10.3389/fphys.2020.00230. eCollection 2020. Front Physiol. 2020. PMID: 32256387 Free PMC article.
-
The transmembrane protein 16A Ca(2+)-activated Cl- channel in airway smooth muscle contributes to airway hyperresponsiveness.Am J Respir Crit Care Med. 2013 Feb 15;187(4):374-81. doi: 10.1164/rccm.201207-1303OC. Epub 2012 Dec 13. Am J Respir Crit Care Med. 2013. PMID: 23239156 Free PMC article.
-
IP3R1 underlies diastolic ANO1 activation and pressure-dependent chronotropy in lymphatic collecting vessels.J Gen Physiol. 2023 Dec 4;155(12):e202313358. doi: 10.1085/jgp.202313358. Epub 2023 Oct 18. J Gen Physiol. 2023. PMID: 37851027 Free PMC article.
-
Activation of the Cl- channel ANO1 by localized calcium signals in nociceptive sensory neurons requires coupling with the IP3 receptor.Sci Signal. 2013 Aug 27;6(290):ra73. doi: 10.1126/scisignal.2004184. Sci Signal. 2013. PMID: 23982204 Free PMC article.
-
Molecular mechanisms of activation and regulation of ANO1-Encoded Ca2+-Activated Cl- channels.Channels (Austin). 2021 Dec;15(1):569-603. doi: 10.1080/19336950.2021.1975411. Channels (Austin). 2021. PMID: 34488544 Free PMC article. Review.
References
-
- Amberg, G.C., and L.F. Santana. 2003. Downregulation of the BK channel β1 subunit in genetic hypertension. Circ. Res. 93:965–971. - PubMed
-
- Becker, P.L., J.J. Singer, J.V. Walsh Jr., and F.S. Fay. 1989. Regulation of calcium concentration in voltage-clamped smooth muscle cells. Science. 244:211–214. - PubMed
-
- Berkefeld, H., C.A. Sailer, W. Bildl, V. Rohde, J.O. Thumfart, S. Eble, N. Klugbauer, E. Reisinger, J. Bischofberger, D. Oliver, et al. 2006. BKCa-Cav channel complexes mediate rapid and localized Ca2+-activated K+ signaling. Science. 314:615–620. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

