Angiotensin II type 1 receptor blocker ameliorates uncoupled endothelial nitric oxide synthase in rats with experimental diabetic nephropathy
- PMID: 18596126
- PMCID: PMC2639062
- DOI: 10.1093/ndt/gfn357
Angiotensin II type 1 receptor blocker ameliorates uncoupled endothelial nitric oxide synthase in rats with experimental diabetic nephropathy
Abstract
Background: Recent studies showed that angiotensin II type 1 receptor blocker (ARB) slows progression of chronic renal disease in patients with type 2 diabetes, regardless of changes in blood pressure. We showed that the imbalance of nitric oxide (NO) and reactive oxygen species (ROS) due to endothelial NO synthase (eNOS) uncoupling contributed to renal dysfunction in the diabetic nephropathy. The aim of this study was to determine the effects of ARB on uncoupled eNOS in rat diabetic nephropathy.
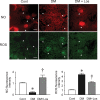
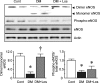
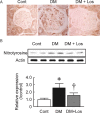
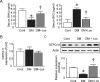
Methods: Diabetes was induced in Sprague-Dawley rats with streptozotocin (65 mg/ kg body weight). After 6 weeks, rats were divided into saline (DM; n = 11) and ARB, losartan groups (DM+Los; n = 11). After 2-week treatment, glomerular ROS production was assessed by 2',7'-dichlorofluorescin diacetate (DCFH-DA)-derived chemiluminescence. Renal NO and ROS production were imaged by confocal laser microscopy after renal perfusion with DCFH-DA and diaminorhodamine-4M acetoxymethyl ester with L-arginine. The dimeric form of eNOS was measured by low-temperature sodium dodecyl sulfate-polyacrylamide gel electrophoresis. Serum tetrahydrobiopterin (BH4) concentrations were determined by high-performance liquid chromatography. Protein and mRNA expression of GTP cyclohydrolase 1 (GTPCH1), key enzyme of BH4 synthesis, were examined.
Results: Losartan attenuated glomerular ROS production in DM. Accelerated ROS production and diminished bioavailable NO caused by NOS uncoupling were noted in DM glomeruli. Losartan reversed the decreased GTPCH1 and decreased dimeric form of eNOS and glomerular NO production by increased BH4 bioavailability.
Conclusions: ARB improved the NOS uncoupling in diabetic nephropathy by increasing BH4 bioavailability.
Figures

References
-
- Guzik TJ, Mussa S, Gastaldi D, et al. Mechanisms of increased vascular superoxide production in human diabetes mellitus: role of NAD(P)H oxidase and endothelial nitric oxide synthase. Circulation. 2002;105:1656–1662. - PubMed
-
- Satoh M, Fujimoto S, Haruna Y, et al. NAD(P)H oxidase and uncoupled nitric oxide synthase are major sources of glomerular superoxide in rats with experimental diabetic nephropathy. Am J Physiol Renal Physiol. 2005;288:F1144–F1152. - PubMed
-
- Brenner BM, Cooper ME, de Zeeuw D, et al. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med. 2001;345:861–869. - PubMed
-
- Parving HH, Lehnert H, Brochner-Mortensen J, et al. The effect of irbesartan on the development of diabetic nephropathy in patients with type 2 diabetes. N Engl J Med. 2001;345:870–878. - PubMed
-
- Malik RA, Schofield IJ, Izzard A, et al. Effects of angiotensin type-1 receptor antagonism on small artery function in patients with type 2 diabetes mellitus. Hypertension. 2005;45:264–269. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

