UNC-18 promotes both the anterograde trafficking and synaptic function of syntaxin
- PMID: 18596236
- PMCID: PMC2526711
- DOI: 10.1091/mbc.e08-02-0160
UNC-18 promotes both the anterograde trafficking and synaptic function of syntaxin
Abstract
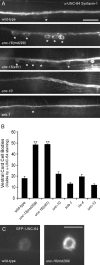
The SM protein UNC-18 has been proposed to regulate several aspects of secretion, including synaptic vesicle docking, priming, and fusion. Here, we show that UNC-18 has a chaperone function in neurons, promoting anterograde transport of the plasma membrane soluble N-ethylmaleimide-sensitive factor attachment protein receptor (SNARE) protein Syntaxin-1. In unc-18 mutants, UNC-64 (Caenorhabditis elegans Syntaxin-1) accumulates in neuronal cell bodies. Colocalization studies and analysis of carbohydrate modifications both suggest that this accumulation occurs in the endoplasmic reticulum. This trafficking defect is specific for UNC-64 Syntaxin-1, because 14 other SNARE proteins and two active zone markers were unaffected. UNC-18 binds to Syntaxin through at least two mechanisms: binding to closed Syntaxin, or to the N terminus of Syntaxin. It is unclear which of these binding modes mediates UNC-18 function in neurons. The chaperone function of UNC-18 was eliminated in double mutants predicted to disrupt both modes of Syntaxin binding, but it was unaffected in single mutants. By contrast, mutations predicted to disrupt UNC-18 binding to the N terminus of Syntaxin caused significant defects in locomotion behavior and responsiveness to cholinesterase inhibitors. Collectively, these results demonstrate the UNC-18 acts as a molecular chaperone for Syntaxin transport in neurons and that the two modes of UNC-18 binding to Syntaxin are involved in different aspects of UNC-18 function.
Figures






References
-
- Advani R. J., Bae H. R., Bock J. B., Chao D. S., Doung Y. C., Prekeris R., Yoo J. S., Scheller R. H. Seven novel mammalian SNARE proteins localize to distinct membrane compartments. J. Biol. Chem. 1998;273:10317–10324. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

