The DEAD-box RNA helicase DDX3 associates with export messenger ribonucleoproteins as well as tip-associated protein and participates in translational control
- PMID: 18596238
- PMCID: PMC2526709
- DOI: 10.1091/mbc.e07-12-1264
The DEAD-box RNA helicase DDX3 associates with export messenger ribonucleoproteins as well as tip-associated protein and participates in translational control
Abstract
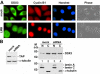
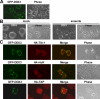
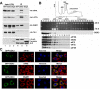
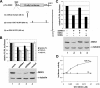
Nuclear export of mRNA is tightly linked to transcription, nuclear mRNA processing, and subsequent maturation in the cytoplasm. Tip-associated protein (TAP) is the major nuclear mRNA export receptor, and it acts coordinately with various factors involved in mRNA expression. We screened for protein factors that associate with TAP and identified several candidates, including RNA helicase DDX3. We demonstrate that DDX3 directly interacts with TAP and that its association with TAP as well as mRNA ribonucleoprotein complexes may occur in the nucleus. Depletion of TAP resulted in nuclear accumulation of DDX3, suggesting that DDX3 is, at least in part, exported along with messenger ribonucleoproteins to the cytoplasm via the TAP-mediated pathway. Moreover, the observation that DDX3 localizes transiently in cytoplasmic stress granules under cell stress conditions suggests a role for DDX3 in translational control. Indeed, DDX3 associates with translation initiation complexes. However, DDX3 is probably not critical for general mRNA translation but may instead promote efficient translation of mRNAs containing a long or structured 5' untranslated region. Given that the DDX3 RNA helicase activity is essential for its involvement in translation, we suggest that DDX3 facilitates translation by resolving secondary structures of the 5'-untranslated region in mRNAs during ribosome scanning.
Figures



References
-
- Berthelot K., Muldoon M., Rajkowitsch L., Hughes J., McCarthy J.E.G. Dynamics and processivity of 40S ribosome scanning on mRNA in yeast. Mol. Microbiol. 2004;51:987–1001. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

