Role of IGF-1/IGF-1R in regulation of invasion in DU145 prostate cancer cells
- PMID: 18598360
- PMCID: PMC2491598
- DOI: 10.1186/1475-2867-8-10
Role of IGF-1/IGF-1R in regulation of invasion in DU145 prostate cancer cells
Abstract
Background: Prostate cancer progression to androgen independence is the primary cause of mortality by this tumor type. The IGF-1/IGF-1R axis is well known to contribute to prostate cancer initiation, but its contribution to invasiveness and the downstream signalling mechanisms that are involved are unclear at present.
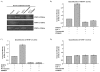
Results: We examined the invasive response of androgen independent DU145 prostate carcinoma cells to IGF-1 stimulation using Matrigel assays. We then examined the signaling mechanisms and protease activities that are associated with this response. IGF-1 significantly increased the invasive capacity of DU145 cells in vitro, and this increase was inhibited by blocking IGF-1R. We further demonstrated that specific inhibitors of the MAPK and PI3-K pathways decrease IGF-1-mediated invasion. To determine potential molecular mechanisms for this change in invasive capacity, we examined changes in expression and activity of matrix metalloproteinases. We observed that IGF-1 increases the enzymatic activity of MMP-2 and MMP-9 in DU145 cells. These changes in activity are due to differences in expression in the case of MMP-9 but not in the case of MMP-2. This observation is corroborated by the fact that correlated changes of expression in a regulator of MMP-2, TIMP-2, were also seen.
Conclusion: This work identifies a specific effect of IGF-1 on the invasive capacity of DU145 prostate cancer cells, and furthermore delineates mechanisms that contribute to this effect.
Figures


Similar articles
-
Differential localization of MT1-MMP in human prostate cancer tissue: role of IGF-1R in MT1-MMP expression.Prostate. 2008 Apr 1;68(5):463-76. doi: 10.1002/pros.20718. Prostate. 2008. PMID: 18196535
-
Insulin-like growth factor-I receptor signalling and acquired resistance to gefitinib (ZD1839; Iressa) in human breast and prostate cancer cells.Endocr Relat Cancer. 2004 Dec;11(4):793-814. doi: 10.1677/erc.1.00799. Endocr Relat Cancer. 2004. PMID: 15613453
-
Crude extract of Euphorbia formosana inhibits the migration and invasion of DU145 human prostate cancer cells: The role of matrix metalloproteinase-2/9 inhibition via the MAPK signaling pathway.Mol Med Rep. 2013 May;7(5):1403-8. doi: 10.3892/mmr.2013.1380. Epub 2013 Mar 20. Mol Med Rep. 2013. PMID: 23525212
-
Proanthocyanidins from grape seeds inhibit expression of matrix metalloproteinases in human prostate carcinoma cells, which is associated with the inhibition of activation of MAPK and NF kappa B.Carcinogenesis. 2004 Jun;25(6):987-95. doi: 10.1093/carcin/bgh095. Epub 2004 Jan 23. Carcinogenesis. 2004. Retraction in: Carcinogenesis. 2012 May;33(5):1121. doi: 10.1093/carcin/bgs146. PMID: 14742313 Retracted.
-
The role of the IGF-I receptor in the regulation of matrix metalloproteinases, tumor invasion and metastasis.Horm Metab Res. 2003 Nov-Dec;35(11-12):802-8. doi: 10.1055/s-2004-814143. Horm Metab Res. 2003. PMID: 14710361 Review.
Cited by
-
IGF-I Signaling in Osterix-Expressing Cells Regulates Secondary Ossification Center Formation, Growth Plate Maturation, and Metaphyseal Formation During Postnatal Bone Development.J Bone Miner Res. 2015 Dec;30(12):2239-48. doi: 10.1002/jbmr.2563. Epub 2015 Jul 29. J Bone Miner Res. 2015. PMID: 26011431 Free PMC article.
-
Inhibition of Insulin-like Growth Factor 1 Receptor/Insulin Receptor Signaling by Small-Molecule Inhibitor BMS-754807 Leads to Improved Survival in Experimental Esophageal Adenocarcinoma.Cancers (Basel). 2024 Sep 17;16(18):3175. doi: 10.3390/cancers16183175. Cancers (Basel). 2024. PMID: 39335147 Free PMC article.
-
MicroRNA-503 acts as a tumor suppressor in glioblastoma for multiple antitumor effects by targeting IGF-1R.Oncol Rep. 2014 Mar;31(3):1445-52. doi: 10.3892/or.2013.2951. Epub 2013 Dec 30. Oncol Rep. 2014. PMID: 24378652 Free PMC article.
-
The Physiological Role of Boron on Health.Biol Trace Elem Res. 2018 Nov;186(1):31-51. doi: 10.1007/s12011-018-1284-3. Epub 2018 Mar 15. Biol Trace Elem Res. 2018. PMID: 29546541 Review.
-
Head and neck tumor cell radiation response occurs in the presence of IGF1.J Dent Res. 2011 Mar;90(3):347-52. doi: 10.1177/0022034510388037. Epub 2010 Nov 12. J Dent Res. 2011. PMID: 21076120 Free PMC article.
References
-
- Meinbach DS, Lokeshwar BL. Insulin-like growth factors and their binding proteins in prostate cancer: cause or consequence? Urol Oncol. 2006;24:294–306. - PubMed
-
- Dunn SE, Ehrlich M, Sharp NJ, Reiss K, Solomon G, Hawkins R, Baserga R, Barrett JC. A dominant negative mutant of the insulin-like growth factor-I receptor inhibits the adhesion, invasion, and metastasis of breast cancer. Cancer Res. 1998;58:3353–3361. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

