Expression-based screening identifies the combination of histone deacetylase inhibitors and retinoids for neuroblastoma differentiation
- PMID: 18607002
- PMCID: PMC2474517
- DOI: 10.1073/pnas.0710413105
Expression-based screening identifies the combination of histone deacetylase inhibitors and retinoids for neuroblastoma differentiation
Abstract
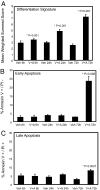
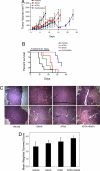
The discovery of new small molecules and their testing in rational combination poses an ongoing problem for rare diseases, in particular, for pediatric cancers such as neuroblastoma. Despite maximal cytotoxic therapy with double autologous stem cell transplantation, outcome remains poor for children with high-stage disease. Because differentiation is aberrant in this malignancy, compounds that modulate transcription, such as histone deacetylase (HDAC) inhibitors, are of particular interest. However, as single agents, HDAC inhibitors have had limited efficacy. In the present study, we use an HDAC inhibitor as an enhancer to screen a small-molecule library for compounds inducing neuroblastoma maturation. To quantify differentiation, we use an enabling gene expression-based screening strategy. The top hit identified in the screen was all-trans-retinoic acid. Secondary assays confirmed greater neuroblastoma differentiation with the combination of an HDAC inhibitor and a retinoid versus either alone. Furthermore, effects of combination therapy were synergistic with respect to inhibition of cellular viability and induction of apoptosis. In a xenograft model of neuroblastoma, animals treated with combination therapy had the longest survival. This work suggests that testing of an HDAC inhibitor and retinoid in combination is warranted for children with neuroblastoma and demonstrates the success of a signature-based screening approach to prioritize compound combinations for testing in rare diseases.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Inhibitors of histone deacetylase (HDAC) restore the p53 pathway in neuroblastoma cells.Br J Pharmacol. 2008 Feb;153(4):657-68. doi: 10.1038/sj.bjp.0707608. Epub 2007 Dec 3. Br J Pharmacol. 2008. PMID: 18059320 Free PMC article.
-
The histone deacetylase inhibitor, CBHA, inhibits growth of human neuroblastoma xenografts in vivo, alone and synergistically with all-trans retinoic acid.Cancer Res. 2001 May 1;61(9):3591-4. Cancer Res. 2001. PMID: 11325825
-
The HDAC6/8/10 inhibitor TH34 induces DNA damage-mediated cell death in human high-grade neuroblastoma cell lines.Arch Toxicol. 2018 Aug;92(8):2649-2664. doi: 10.1007/s00204-018-2234-8. Epub 2018 Jun 9. Arch Toxicol. 2018. PMID: 29947893 Free PMC article.
-
Histone deacetylase inhibitors: mechanisms of cell death and promise in combination cancer therapy.Cancer Lett. 2008 Sep 28;269(1):7-17. doi: 10.1016/j.canlet.2008.03.037. Epub 2008 May 6. Cancer Lett. 2008. PMID: 18462867 Review.
-
Targeting histone deacetylases in neuroblastoma.Curr Pharm Des. 2009;15(4):436-47. doi: 10.2174/138161209787315774. Curr Pharm Des. 2009. PMID: 19199971 Review.
Cited by
-
Anti-GD2 mAb and Vorinostat synergize in the treatment of neuroblastoma.Oncoimmunology. 2016 Mar 28;5(6):e1164919. doi: 10.1080/2162402X.2016.1164919. eCollection 2016 Jun. Oncoimmunology. 2016. PMID: 27471639 Free PMC article.
-
Type III TGF-β receptor promotes FGF2-mediated neuronal differentiation in neuroblastoma.J Clin Invest. 2013 Nov;123(11):4786-98. doi: 10.1172/JCI69657. J Clin Invest. 2013. PMID: 24216509 Free PMC article.
-
Discovery of 5-sulfonyltetrazoles as neuroblastoma differentiation agents.Bioorg Med Chem Lett. 2023 Oct 1;94:129455. doi: 10.1016/j.bmcl.2023.129455. Epub 2023 Aug 18. Bioorg Med Chem Lett. 2023. PMID: 37597697 Free PMC article.
-
DNER, an epigenetically modulated gene, regulates glioblastoma-derived neurosphere cell differentiation and tumor propagation.Stem Cells. 2009 Jul;27(7):1473-86. doi: 10.1002/stem.89. Stem Cells. 2009. PMID: 19544453 Free PMC article.
-
Identification of a novel gene signature for neuroblastoma differentiation using a Boolean implication network.Genes Chromosomes Cancer. 2023 Jun;62(6):313-331. doi: 10.1002/gcc.23124. Epub 2023 Feb 13. Genes Chromosomes Cancer. 2023. PMID: 36680522 Free PMC article.
References
-
- George RE, et al. High-risk neuroblastoma treated with tandem autologous peripheral-blood stem cell-supported transplantation: Long-term survival update. J Clin Oncol. 2006;24:2891–2896. - PubMed
-
- Furchert SE. Inhibitors of histone deacetylases as potential therapeutic tools for high-risk embryonal tumors of the nervous system of childhood. Int J Cancer. 2007;120:1787–1794. - PubMed
-
- Bolden JE, Peart MJ, Johnstone RW. Anticancer activities of histone deacetylase inhibitors. Nature Rev. 2006;5:769–784. - PubMed
-
- Matthay KK, et al. Treatment of high-risk neuroblastoma with intensive chemotherapy, radiotherapy, autologous bone marrow transplantation, and 13-cis-retinoic acid. Children's Cancer Group. N Engl J Med. 1999;341:1165–1173. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

