Distinct role of long 3' UTR BDNF mRNA in spine morphology and synaptic plasticity in hippocampal neurons
- PMID: 18614020
- PMCID: PMC2527207
- DOI: 10.1016/j.cell.2008.05.045
Distinct role of long 3' UTR BDNF mRNA in spine morphology and synaptic plasticity in hippocampal neurons
Abstract
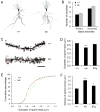
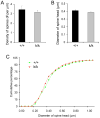
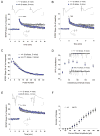
The brain produces two brain-derived neurotrophic factor (BDNF) transcripts, with either short or long 3' untranslated regions (3' UTRs). The physiological significance of the two forms of mRNAs encoding the same protein is unknown. Here, we show that the short and long 3' UTR BDNF mRNAs are involved in different cellular functions. The short 3' UTR mRNAs are restricted to somata, whereas the long 3' UTR mRNAs are also localized in dendrites. In a mouse mutant where the long 3' UTR is truncated, dendritic targeting of BDNF mRNAs is impaired. There is little BDNF in hippocampal dendrites despite normal levels of total BDNF protein. This mutant exhibits deficits in pruning and enlargement of dendritic spines, as well as selective impairment in long-term potentiation in dendrites, but not somata, of hippocampal neurons. These results provide insights into local and dendritic actions of BDNF and reveal a mechanism for differential regulation of subcellular functions of proteins.
Figures




References
-
- Aakalu G, Smith WB, Nguyen N, Jiang C, Schuman EM. Dynamic visualization of local protein synthesis in hippocampal neurons. Neuron. 2001;30:489–502. - PubMed
-
- Blichenberg A, Rehbein M, Muller R, Garner CC, Richter D, Kindler S. Identification of a cis-acting dendritic targeting element in the mRNA encoding the alpha subunit of Ca2+/calmodulin-dependent protein kinase II. Eur J Neurosci. 2001;13:1881–1888. - PubMed
-
- Bramham CR, Wells DG. Dendritic mRNA: transport, translation and function. Nat Rev Neurosci. 2007;8:776–789. - PubMed
-
- Derkach VA, Oh MC, Guire ES, Soderling TR. Regulatory mechanisms of AMPA receptors in synaptic plasticity. Nat Rev Neurosci. 2007;8:101–113. - PubMed
-
- Figurov A, Pozzo-Miller LD, Olafsson P, Wang T, Lu B. Regulation of synaptic responses to high-frequency stimulation and LTP by neurotrophins in the hippocampus. Nature. 1996;381:706–709. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

