Phosphorylation of Thr-178 and Thr-184 in the TAK1 T-loop is required for interleukin (IL)-1-mediated optimal NFkappaB and AP-1 activation as well as IL-6 gene expression
- PMID: 18617512
- PMCID: PMC2528992
- DOI: 10.1074/jbc.M802825200
Phosphorylation of Thr-178 and Thr-184 in the TAK1 T-loop is required for interleukin (IL)-1-mediated optimal NFkappaB and AP-1 activation as well as IL-6 gene expression
Abstract
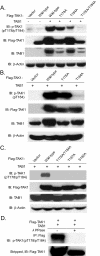
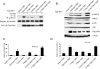
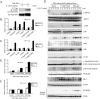
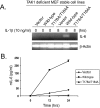
TAK1 (transforming growth factor-beta-activated kinase 1), a mitogen-activated protein kinase kinase kinase, is activated by various cytokines, including interleukin-1 (IL-1). However, the precise regulation for TAK1 activation at the molecular level is still not fully understood. Here we report that dual phosphorylation of Thr-178 and Thr-184 residues within the kinase activation loop of TAK1 is essential for TAK1-mediated NFkappaB and AP-1 activation. Once co-overexpressed with TAB1, TAK1 mutant with alanine substitution of these two residues fails to activate IKKbeta-mediated NFkappaB and JNK-mediated AP-1, whereas TAK1 mutant with replacement of these two sites with acidic residues acts like the TAK1 wild type. Consistently, TAK1 mutant with alanine substitution of these two residues severely inhibits IL-1-induced NFkappaB and AP-1 activities, whereas TAK1 mutant with replacement of these two sites with acidic residues slightly enhances IL-1-induced NFkappaB and AP-1 activities compared with the TAK1 wild-type. IL-1 induces the phosphorylation of endogenous TAK1 at Thr-178 and Thr-184. Reconstitution of TAK1-deficient mouse embryo fibroblast cells with wild-type TAK1 or a TAK1 mutant containing threonine 178 and 184 to alanine mutations revealed the importance of these two sites in IL-1-mediated IKK-NFkappaB and JNK-AP-1 activation as well as IL-1-induced IL-6 gene expression. Our finding is the first report that substitution of key serine/threonine residues with acidic residues mimics the phosphorylated state of TAK1 and renders TAK1 active during its induced activation.
Figures
Similar articles
-
Lysine 63-linked polyubiquitination of TAK1 at lysine 158 is required for tumor necrosis factor alpha- and interleukin-1beta-induced IKK/NF-kappaB and JNK/AP-1 activation.J Biol Chem. 2010 Feb 19;285(8):5347-60. doi: 10.1074/jbc.M109.076976. Epub 2009 Dec 28. J Biol Chem. 2010. PMID: 20038579 Free PMC article.
-
TAK1 lysine 158 is required for TGF-β-induced TRAF6-mediated Smad-independent IKK/NF-κB and JNK/AP-1 activation.Cell Signal. 2011 Jan;23(1):222-7. doi: 10.1016/j.cellsig.2010.09.006. Epub 2010 Sep 16. Cell Signal. 2011. PMID: 20837137 Free PMC article.
-
TAB4 stimulates TAK1-TAB1 phosphorylation and binds polyubiquitin to direct signaling to NF-kappaB.J Biol Chem. 2008 Jul 11;283(28):19245-54. doi: 10.1074/jbc.M800943200. Epub 2008 May 2. J Biol Chem. 2008. PMID: 18456659 Free PMC article.
-
Roles for TAB1 in regulating the IL-1-dependent phosphorylation of the TAB3 regulatory subunit and activity of the TAK1 complex.Biochem J. 2008 Feb 1;409(3):711-22. doi: 10.1042/BJ20071149. Biochem J. 2008. PMID: 18021073
-
TAK1 mitogen-activated protein kinase kinase kinase is activated by autophosphorylation within its activation loop.J Biol Chem. 2000 Mar 10;275(10):7359-64. doi: 10.1074/jbc.275.10.7359. J Biol Chem. 2000. PMID: 10702308
Cited by
-
Targeting of TGF-β-activated protein kinase 1 inhibits chemokine (C-C motif) receptor 7 expression, tumor growth and metastasis in breast cancer.Oncotarget. 2015 Jan 20;6(2):995-1007. doi: 10.18632/oncotarget.2739. Oncotarget. 2015. PMID: 25557171 Free PMC article.
-
Transforming growth factor β activated kinase 1: a potential therapeutic target for rheumatic diseases.Rheumatology (Oxford). 2017 Jul 1;56(7):1060-1068. doi: 10.1093/rheumatology/kew301. Rheumatology (Oxford). 2017. PMID: 27550296 Free PMC article. Review.
-
Autoactivation of transforming growth factor beta-activated kinase 1 is a sequential bimolecular process.J Biol Chem. 2010 Aug 13;285(33):25753-66. doi: 10.1074/jbc.M109.093468. Epub 2010 Jun 10. J Biol Chem. 2010. PMID: 20538596 Free PMC article.
-
Linking JNK Activity to the DNA Damage Response.Genes Cancer. 2013 Sep;4(9-10):360-8. doi: 10.1177/1947601913486347. Genes Cancer. 2013. PMID: 24349633 Free PMC article. Review.
-
Engineering allosteric regulation in protein kinases.Sci Signal. 2018 Nov 6;11(555):eaar3250. doi: 10.1126/scisignal.aar3250. Sci Signal. 2018. PMID: 30401787 Free PMC article.
References
-
- Dinarello, C. A. (1997) Cytokine Growth Factor Rev. 8 253–265 - PubMed
-
- O'Neill, L. A., and Greene, C. (1998) J. Leukocyte Biol. 63 650–657 - PubMed
-
- Arend, W. P. (2002) Cytokine Growth Factor Rev. 13 323–340 - PubMed
-
- Beutler, B. (2004) Nature 430 257–263 - PubMed
-
- Baud, V., and Karin, M. (2001) Trends Cell Biol. 11 372–377 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

