Biosynthesis of a rare di-N-acetylated sugar in the lipopolysaccharides of both Pseudomonas aeruginosa and Bordetella pertussis occurs via an identical scheme despite different gene clusters
- PMID: 18621892
- PMCID: PMC2546786
- DOI: 10.1128/JB.00579-08
Biosynthesis of a rare di-N-acetylated sugar in the lipopolysaccharides of both Pseudomonas aeruginosa and Bordetella pertussis occurs via an identical scheme despite different gene clusters
Abstract
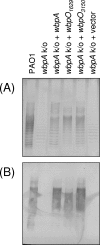
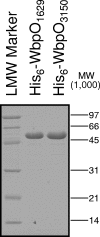
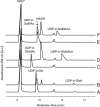
Pseudomonas aeruginosa and Bordetella pertussis produce lipopolysaccharide (LPS) that contains 2,3-diacetamido-2,3-dideoxy-D-mannuronic acid (D-ManNAc3NAcA). A five-enzyme biosynthetic pathway that requires WbpA, WbpB, WbpE, WbpD, and WbpI has been proposed for the production of this sugar in P. aeruginosa, based on analysis of genes present in the B-band LPS biosynthesis cluster. In the analogous B. pertussis cluster, homologs of wbpB to wbpI were present, but a putative dehydrogenase gene was missing; therefore, the biosynthetic mechanism for UDP-D-ManNAc3NAcA was unclear. Nonpolar knockout mutants of each P. aeruginosa gene were constructed. Complementation analysis of the mutants demonstrated that B-band LPS production was restored to P. aeruginosa knockout mutants when the relevant B. pertussis genes were supplied in trans. Thus, the genes that encode the putative oxidase, transaminase, N-acetyltransferase, and epimerase enzymes in B. pertussis are functional homologs of those in P. aeruginosa. Two candidate dehydrogenase genes were located by searching the B. pertussis genome; these have 80% identity to P. aeruginosa wbpO (serotype O6) and 32% identity to wbpA (serotype O5). These genes, wbpO(1629) and wbpO(3150), were shown to complement a wbpA knockout of P. aeruginosa. Capillary electrophoresis was used to characterize the enzymatic activities of purified WbpO(1629) and WbpO(3150), and mass spectrometry analysis confirmed that the two enzymes are dehydrogenases capable of converting UDP-D-GlcNAc, UDP-D-GalNAc, to a lesser extent, and UDP-D-Glc, to a much lesser extent. Together, these results suggest that B. pertussis produces UDP-D-ManNAc3NAcA through the same pathway proposed for P. aeruginosa, despite differences in the genomic context of the genes involved.
Figures



Similar articles
-
Characterization of WbpB, WbpE, and WbpD and reconstitution of a pathway for the biosynthesis of UDP-2,3-diacetamido-2,3-dideoxy-D-mannuronic acid in Pseudomonas aeruginosa.J Biol Chem. 2009 May 1;284(18):11854-62. doi: 10.1074/jbc.M808583200. Epub 2009 Mar 12. J Biol Chem. 2009. PMID: 19282284 Free PMC article.
-
Biosynthesis of UDP-GlcNAc(3NAc)A by WbpB, WbpE, and WbpD: enzymes in the Wbp pathway responsible for O-antigen assembly in Pseudomonas aeruginosa PAO1.Biochemistry. 2009 Jun 16;48(23):5446-55. doi: 10.1021/bi900186u. Biochemistry. 2009. PMID: 19348502 Free PMC article.
-
Identification and biochemical characterization of two novel UDP-2,3-diacetamido-2,3-dideoxy-alpha-D-glucuronic acid 2-epimerases from respiratory pathogens.Biochem J. 2007 Jul 1;405(1):123-30. doi: 10.1042/BJ20070017. Biochem J. 2007. PMID: 17346239 Free PMC article.
-
Review: Lipopolysaccharide biosynthesis in Pseudomonas aeruginosa.Innate Immun. 2009 Oct;15(5):261-312. doi: 10.1177/1753425909106436. Epub 2009 Aug 26. Innate Immun. 2009. PMID: 19710102 Review.
-
Genetics of O-antigen biosynthesis in Pseudomonas aeruginosa.Microbiol Mol Biol Rev. 1999 Sep;63(3):523-53. doi: 10.1128/MMBR.63.3.523-553.1999. Microbiol Mol Biol Rev. 1999. PMID: 10477307 Free PMC article. Review.
Cited by
-
Structural and functional studies of WlbA: A dehydrogenase involved in the biosynthesis of 2,3-diacetamido-2,3-dideoxy-D-mannuronic acid.Biochemistry. 2010 Sep 14;49(36):7939-48. doi: 10.1021/bi101103s. Biochemistry. 2010. PMID: 20690587 Free PMC article.
-
Structural investigation on WlaRG from Campylobacter jejuni: A sugar aminotransferase.Protein Sci. 2017 Mar;26(3):586-599. doi: 10.1002/pro.3109. Epub 2017 Feb 9. Protein Sci. 2017. PMID: 28028852 Free PMC article.
-
Genetic and Functional Diversity of Pseudomonas aeruginosa Lipopolysaccharide.Front Microbiol. 2011 Jun 1;2:118. doi: 10.3389/fmicb.2011.00118. eCollection 2011. Front Microbiol. 2011. PMID: 21687428 Free PMC article.
-
Molecular structure of WlbB, a bacterial N-acetyltransferase involved in the biosynthesis of 2,3-diacetamido-2,3-dideoxy-D-mannuronic acid.Biochemistry. 2010 Jun 8;49(22):4644-53. doi: 10.1021/bi1005738. Biochemistry. 2010. PMID: 20433200 Free PMC article.
-
Characterization of WbpB, WbpE, and WbpD and reconstitution of a pathway for the biosynthesis of UDP-2,3-diacetamido-2,3-dideoxy-D-mannuronic acid in Pseudomonas aeruginosa.J Biol Chem. 2009 May 1;284(18):11854-62. doi: 10.1074/jbc.M808583200. Epub 2009 Mar 12. J Biol Chem. 2009. PMID: 19282284 Free PMC article.
References
-
- Allen, A., and D. Maskell. 1996. The identification, cloning and mutagenesis of a genetic locus required for lipopolysaccharide biosynthesis in Bordetella pertussis. Mol. Microbiol. 1937-52. - PubMed
-
- Bélanger, M., L. L. Burrows, and J. S. Lam. 1999. Functional analysis of genes responsible for the synthesis of the B-band O antigen of Pseudomonas aeruginosa serotype O6 lipopolysaccharide. Microbiology 1453505-3521. - PubMed
-
- Burrows, L. L., D. F. Charter, and J. S. Lam. 1996. Molecular characterization of the Pseudomonas aeruginosa serotype O5 (PAO1) B-band lipopolysaccharide gene cluster. Mol. Microbiol. 22481-495. - PubMed
-
- Burrows, L. L., K. E. Pigeon, and J. S. Lam. 2000. Pseudomonas aeruginosa B-band lipopolysaccharide genes wbpA and wbpI and their Escherichia coli homologues wecC and wecB are not functionally interchangeable. FEMS Microbiol. Lett. 189135-141. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

