Randomized phase III clinical trial designs for targeted agents
- PMID: 18628448
- PMCID: PMC2569946
- DOI: 10.1158/1078-0432.CCR-08-0288
Randomized phase III clinical trial designs for targeted agents
Abstract
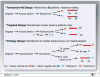
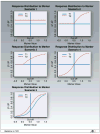
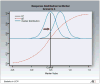
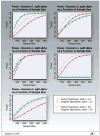
Purpose: Cancer therapies with mechanisms of action which are very different from the more conventional chemotherapies are now being developed. In this article, we investigate the performance of several phase III clinical trial designs, both for testing the overall efficacy of a targeted agent and for testing its efficacy in a subgroup of patients with a tumor marker present. We study different designs and different underlying scenarios assuming continuous markers, and assess the trade-off between the number of patients on the study and the effectiveness of treatment in the subgroup of marker-positive patients.
Experimental design: We investigate binary outcomes and use simulation studies to determine sample size and power for the different designs and the various scenarios. We also simulate marker prevalence and marker misclassification and evaluate their effect on power and sample size.
Results: In general, a targeted design which randomizes patients with the appropriate marker status performs the best in all scenarios with an underlying true predictive marker. Randomizing all patients regardless of their marker values performs as well as or better in most cases than a clinical trial that randomizes the patient to a treatment strategy based on marker value versus standard of care.
Conclusion: If there is the possibility that the new treatment helps marker-negative patients, or that the cutpoint determining marker status has not been well established and the marker prevalence is large enough, we recommend randomizing all patients regardless of marker values, but using a design such that both the overall and the targeted subgroup hypothesis can be tested.
Conflict of interest statement
Figures



References
-
- Shepherd FA, Pereira J, Ciuleanu TE, et al. Erlotinib in previously treated non-small-cell lung cancer. N Engl J Med. 2005;353:123–32. - PubMed
-
- Hirsch FR, Varella-Garcia M, Bunn PA, et al. Epidermal growth factor receptor in non-small-cell lung carcinomas: correlation between gene copy number and protein expression and impact on prognosis. J Clin Oncol. 2003;21:3798–807. - PubMed
-
- Menendez JA, Mehmi I, Lupu R. Trastuzumab in combination with heregulin-activated Her-2 (erbB-2) triggers a receptor-enhanced chemosensitivity effect in the absence of Her-2 overexpression. J Clin Oncol. 2006;24:3735–46. - PubMed
-
- Heinrich MC, Corless CL, Demetri GD, et al. Kinase mutations and imatinib response in patients with metastatic gastrointestinal stromal tumor. J Clin Oncol. 2003;21:4342–9. - PubMed
-
- Hoering A, Crowley J. Clinical trial designs for multiple myeloma. Clin Adv Hematol. 2007;5:309–16. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

