On the nature of partial agonism in the nicotinic receptor superfamily
- PMID: 18633353
- PMCID: PMC2629928
- DOI: 10.1038/nature07139
On the nature of partial agonism in the nicotinic receptor superfamily
Abstract
Partial agonists are ligands that bind to receptors but produce only a small maximum response even at concentrations where all receptors are occupied. In the case of ligand-activated ion channels, it has been supposed since 1957 that partial agonists evoke a small response because they are inefficient at eliciting the change of conformation between shut and open states of the channel. We have investigated partial agonists for two members of the nicotinic superfamily-the muscle nicotinic acetylcholine receptor and the glycine receptor-and find that the open-shut reaction is similar for both full and partial agonists, but the response to partial agonists is limited by an earlier conformation change ('flipping') that takes place while the channel is still shut. This has implications for the interpretation of structural studies, and in the future, for the design of partial agonists for therapeutic use.
Figures
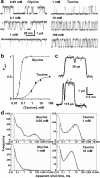
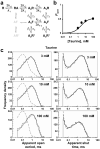
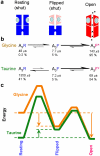
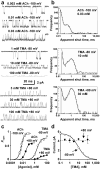
Comment in
-
Pharmacology: Unready for action.Nature. 2008 Aug 7;454(7205):704-5. doi: 10.1038/454704a. Nature. 2008. PMID: 18685692 No abstract available.
References
-
- del Castillo J, Katz B. Interaction at end-plate receptors between different choline derivatives. Proc. R. Soc. Lond. B. 1957;146:369–381. - PubMed
-
- Wyman J, Allen DW. The problem of the heme Interactions in hemoglobin and the basis of the Bohr effect. J. Polym. Sci. 1951;VII:499–518.
-
- Monod J, Wyman J, Changeux J-P. On the nature of allosteric transitions: a plausible model. J. Mol. Biol. 1965;12:88–118. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

