Azaspirene, a fungal product, inhibits angiogenesis by blocking Raf-1 activation
- PMID: 18637013
- PMCID: PMC2687096
- DOI: 10.1111/j.1349-7006.2008.00890.x
Azaspirene, a fungal product, inhibits angiogenesis by blocking Raf-1 activation
Abstract
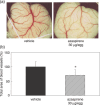
Angiogenesis is an inevitable event in tumor progression and metastasis, and thus has been a compelling target for cancer therapy in recent years. Effective inhibition of tumor progression and metastasis could become a promising way to treat tumor-induced angiogenesis. We discovered that a fungus, Neosartorya sp., isolated from a soil sample, produced a new angiogenesis inhibitor, which we designated azaspirene. Azaspirene was previously shown to inhibit human umbilical vein endothelial cell (HUVEC) migration induced by vascular endothelial growth factor (VEGF) at an effective dose, 100% of 27 micromol/L without significant cell toxicity. In the present study, we investigated the antiangiogenic activity of azaspirene in vivo. Azaspirene treatment reduced the number of tumor-induced blood vessels. Administration of azaspirene at 30 microg/egg resulted in inhibition of angiogenesis (23.6-45.3% maximum inhibition relative to the controls) in a chicken chorioallantoic membrane assay. Next, we elucidated the molecular mechanism of antiangiogenesis of azaspirene. We investigated the effects of azaspirene on VEGF-induced activation of the mitogen-activated protein kinase signaling pathway in HUVEC. In vitro experiments indicated that azaspirene suppressed Raf-1 activation induced by VEGF without affecting the activation of kinase insert domain-containing receptor/fetal liver kinase 1 (VEGF receptor 2). Additionally, azaspirene preferentially inhibited the growth of HUVEC but not that of the non-vascular endothelial cells NIH3T3, HeLa, MSS31, and MCF-7. Taken together, these results demonstrate that azaspirene is a novel inhibitor of angiogenesis and Raf-1 activation that contains a unique carbon skeleton in its molecular structure.
Figures
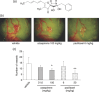




 , NIH3T3 (IC50 = 216 µmol/L);
, NIH3T3 (IC50 = 216 µmol/L);  , HeLa (IC50 = 189 µmol/L);
, HeLa (IC50 = 189 µmol/L);  , MSS31 (IC50 = 173 µmol/L);
, MSS31 (IC50 = 173 µmol/L);  , MCF‐7 (IC50 = 75.6 µmol/L); and
, MCF‐7 (IC50 = 75.6 µmol/L); and  , HUVEC (IC50 = 62.1 µmol/L). Each value is expressed relative to the 1% dimethyl sulfoxide (DMSO) control group; bars, SD. The statistical significance of differences between the growth inhibition (%) of HUVEC with azaspirene at 81 µmol/L was determined using one‐way ANOVA, Tukey method analysis, repeated measures. *P < 0.05; **P < 0.01 was taken as the level of statistical significance. (b) Effects of azaspirene on vascular endothelial growth factor (VEGF)‐induced ERK1 and 2 phosphorylation in HEK293T cells expressing kinase insert domain‐containing receptor/fetal liver kinase 1 (KDR/Flk‐1). HEK293T cells transfected with KDR/Flk‐1 were incubated for 1 h in the absence (DMSO) or presence of various concentrations of azaspirene (27, 81, or 270 µmol/L). These cells were then treated with 50 ng/mL VEGF for 5 min. After stimulation, the cells were harvested, and western blotting was carried out. IB, western blotting analysis. The results shown are representative of three experiments.
, HUVEC (IC50 = 62.1 µmol/L). Each value is expressed relative to the 1% dimethyl sulfoxide (DMSO) control group; bars, SD. The statistical significance of differences between the growth inhibition (%) of HUVEC with azaspirene at 81 µmol/L was determined using one‐way ANOVA, Tukey method analysis, repeated measures. *P < 0.05; **P < 0.01 was taken as the level of statistical significance. (b) Effects of azaspirene on vascular endothelial growth factor (VEGF)‐induced ERK1 and 2 phosphorylation in HEK293T cells expressing kinase insert domain‐containing receptor/fetal liver kinase 1 (KDR/Flk‐1). HEK293T cells transfected with KDR/Flk‐1 were incubated for 1 h in the absence (DMSO) or presence of various concentrations of azaspirene (27, 81, or 270 µmol/L). These cells were then treated with 50 ng/mL VEGF for 5 min. After stimulation, the cells were harvested, and western blotting was carried out. IB, western blotting analysis. The results shown are representative of three experiments.References
-
- Bussolino F, Mantovani A, Persico G. Molecular mechanisms of blood vessel formation. Trends Biochem Sci 1997; 22: 251–6. - PubMed
-
- Hanhan D, Folkman J. Patterns and emerging mechanisms of the angiogenic switch during tumorigenesis. Cell 1996; 82: 353–64. - PubMed
-
- Folkman J. What is the evidence that tumors are angiogenesis dependent? J Natl Cancer Inst 1990; 80: 4–6. - PubMed
-
- Fidler IJ, Ellis LM. The implications of angiogenesis for the biology and therapy of cancer metastasis. Cell 1994; 79: 185–8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

