Volume and neuron number of the lateral geniculate nucleus in schizophrenia and mood disorders
- PMID: 18642008
- PMCID: PMC2832194
- DOI: 10.1007/s00401-008-0410-2
Volume and neuron number of the lateral geniculate nucleus in schizophrenia and mood disorders
Abstract
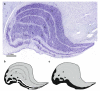
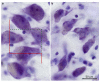
Subjects with schizophrenia show deficits in visual perception that suggest changes predominantly in the magnocellular pathway and/or the dorsal visual stream important for visiospatial perception. We previously found a substantial 25% reduction in neuron number of the primary visual cortex (Brodmann's area 17, BA17) in postmortem tissue from subjects with schizophrenia. Also, many studies have found reduced volume and neuron number of the pulvinar--the large thalamic association nucleus involved in higher-order visual processing. Here, we investigate if the lateral geniculate nucleus (LGN), the visual relay nucleus of the thalamus, has structural changes in schizophrenia. We used stereological methods based on unbiased principles of sampling (Cavalieri's principle and the optical fractionator) to estimate the total volume and neuron number of the magno- and parovocellular parts of the left LGN in postmortem brains from nine subjects with schizophrenia, seven matched normal comparison subjects and 13 subjects with mood disorders. No significant schizophrenia-related structural differences in volume or neuron number of the left LGN or its major subregions were found, but we did observe a significantly increased total volume of the LGN, and of the parvocellular lamina and interlaminar regions, in the mood group. These findings do not support the hypothesis that subjects with schizophrenia have structural changes in the LGN. Therefore, our previous observation of a schizophrenia-related reduction of the primary visual cortex is probably not secondary to a reduction in the LGN.
Figures








Similar articles
-
Stereologic analysis of the lateral geniculate nucleus of the thalamus in normal and schizophrenic subjects.Psychiatry Res. 2007 May 30;151(1-2):1-10. doi: 10.1016/j.psychres.2006.11.003. Epub 2007 Mar 26. Psychiatry Res. 2007. PMID: 17383740 Free PMC article.
-
Primary visual cortex volume and total neuron number are reduced in schizophrenia.J Comp Neurol. 2007 Mar 10;501(2):290-301. doi: 10.1002/cne.21243. J Comp Neurol. 2007. PMID: 17226750
-
Stereological analysis of the mediodorsal thalamic nucleus in schizophrenia: volume, neuron number, and cell types.J Comp Neurol. 2004 May 10;472(4):449-62. doi: 10.1002/cne.20055. J Comp Neurol. 2004. PMID: 15065119
-
Mapping the primate lateral geniculate nucleus: a review of experiments and methods.J Physiol Paris. 2014 Feb;108(1):3-10. doi: 10.1016/j.jphysparis.2013.10.001. Epub 2013 Nov 21. J Physiol Paris. 2014. PMID: 24270042 Free PMC article. Review.
-
Volumes of association thalamic nuclei in schizophrenia: a postmortem study.Schizophr Res. 2003 Apr 1;60(2-3):141-55. doi: 10.1016/s0920-9964(02)00307-9. Schizophr Res. 2003. PMID: 12591578 Review.
Cited by
-
Improving the Quantification of the Lateral Geniculate Nucleus in Magnetic Resonance Imaging Using a Novel 3D-Edge Enhancement Technique.Front Comput Neurosci. 2021 Dec 3;15:708866. doi: 10.3389/fncom.2021.708866. eCollection 2021. Front Comput Neurosci. 2021. PMID: 34924983 Free PMC article.
-
Pyramidal neuron number in layer 3 of primary auditory cortex of subjects with schizophrenia.Brain Res. 2009 Aug 18;1285:42-57. doi: 10.1016/j.brainres.2009.06.019. Epub 2009 Jun 12. Brain Res. 2009. PMID: 19524554 Free PMC article.
-
Immunocytochemical profiles of inferior colliculus neurons in the rat and their changes with aging.Front Neural Circuits. 2012 Sep 21;6:68. doi: 10.3389/fncir.2012.00068. eCollection 2012. Front Neural Circuits. 2012. PMID: 23049499 Free PMC article.
-
The search for true numbers of neurons and glial cells in the human brain: A review of 150 years of cell counting.J Comp Neurol. 2016 Dec 15;524(18):3865-3895. doi: 10.1002/cne.24040. Epub 2016 Jun 16. J Comp Neurol. 2016. PMID: 27187682 Free PMC article. Review.
-
Schizophrenia and cortical blindness: protective effects and implications for language.Front Hum Neurosci. 2014 Nov 28;8:940. doi: 10.3389/fnhum.2014.00940. eCollection 2014. Front Hum Neurosci. 2014. PMID: 25506321 Free PMC article.
References
-
- Abitz M, Nielsen RD, Jones EG, Laursen H, Graem N, Pakkenberg B. Excess of neurons in the human newborn mediodorsal thalamus compared with that of the adult. Cereb Cortex. 2007;17:2573–2578. doi:10.1093/cercor/bhl163. - PubMed
-
- American Psychiatric Association . Diagnostic and statistical manual of mental disorders. 4th edn American Psychiatric Association; Washington, DC: 1994. DSM-IV.
-
- Andersen BB, Gundersen HJG. Pronounced loss of cell nuclei and anisotropic deformation of thick sections. J Microsc. 1999;196:69–73. doi:10.1046/j.1365-2818.1999.00555.x. - PubMed
-
- Andreasen NC, O’Leary DS, Flaum M, Nopoulos P, Watkins GL, Ponto LL Boles, et al. Hypofrontality in schizophrenia: distributed dysfunctional circuits in neuroleptic-naïve patients. Lancet. 1997;349:1730–1734. doi:10.1016/S0140-6736(96)08258-X. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

