The forkhead transcription factor FOXO3a increases phosphoinositide-3 kinase/Akt activity in drug-resistant leukemic cells through induction of PIK3CA expression
- PMID: 18644865
- PMCID: PMC2547013
- DOI: 10.1128/MCB.01265-07
The forkhead transcription factor FOXO3a increases phosphoinositide-3 kinase/Akt activity in drug-resistant leukemic cells through induction of PIK3CA expression
Abstract
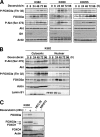
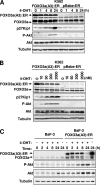
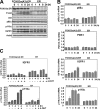
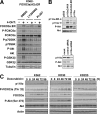
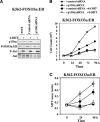
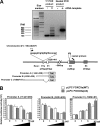
The phosphoinositide-3 kinase (PI3K)/Akt signal pathway plays a key role in the tumorigenesis of many cancers and in the subsequent development of drug resistance. Using the K562 chronic myelogenous leukemia (CML) cell line and the doxorubicin-resistant derivative lines KD30 and KD225 as models, we observed that enhanced PI3K/Akt activity and the acquisition of chemoresistance correlated unexpectedly with the increased expression and nuclear accumulation of FOXO3a. Moreover, we found that the induction of FOXO3a activity in naïve K562 cells was sufficient to enhance PI3K/Akt activity and to confer resistance to the cytotoxic effects of doxorubicin. Conversely, the knockdown of endogenous FOXO3a expression reduced PI3K/Akt activity and sensitized these cells to doxorubicin. Further chromatin immunoprecipitation and promoter mutation analyses demonstrated that FOXO3a regulates the expression of the PI3K catalytic subunit p110alpha through the activation of a promoter region proximal to a novel untranslated exon upstream from the reported transcription start site of the p110alpha gene PIK3CA. As was the case for FOXO3a, the expression or knockdown of p110alpha was sufficient to amplify or reduce PI3K/Akt activity, respectively. Thus, our results suggest that the chronic activation of FOXO3a by doxorubicin in CML cells can enhance survival through a feedback mechanism that involves enhanced p110alpha expression and hyperactivation of the PI3K/Akt pathway.
Figures



References
-
- Birkenkamp, K. U., A. Essafi, K. E. van der Vos, M. da Costa, R. C. Hui, F. Holstege, L. Koenderman, E. W. Lam, and P. J. Coffer. 2007. FOXO3a induces differentiation of Bcr-Abl-transformed cells through transcriptional down-regulation of Id1. J. Biol. Chem. 2822211-2220. - PubMed
-
- Brunet, A., L. B. Sweeney, J. F. Sturgill, K. F. Chua, P. L. Greer, Y. Lin, H. Tran, S. E. Ross, R. Mostoslavsky, H. Y. Cohen, L. S. Hu, H. L. Cheng, M. P. Jedrychowski, S. P. Gygi, D. A. Sinclair, F. W. Alt, and M. E. Greenberg. 2004. Stress-dependent regulation of FOXO transcription factors by the SIRT1 deacetylase. Science 3032011-2015. - PubMed
-
- Charvet, C., I. Alberti, F. Luciano, A. Jacquel, A. Bernard, P. Auberger, and M. Deckert. 2003. Proteolytic regulation of Forkhead transcription factor FOXO3a by caspase-3-like proteases. Oncogene 224557-4568. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous
