Effects of antibiotics on quorum sensing in Pseudomonas aeruginosa
- PMID: 18644954
- PMCID: PMC2565867
- DOI: 10.1128/AAC.01230-07
Effects of antibiotics on quorum sensing in Pseudomonas aeruginosa
Abstract
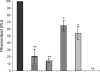
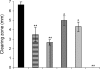
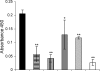
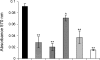
During infection, Pseudomonas aeruginosa employs bacterial communication (quorum sensing [QS]) to coordinate the expression of tissue-damaging factors. QS-controlled gene expression plays a pivotal role in the virulence of P. aeruginosa, and QS-deficient mutants cause less severe infections in animal infection models. Treatment of cystic fibrosis (CF) patients chronically infected with P. aeruginosa with the macrolide antibiotic azithromycin (AZM) has been demonstrated to improve the clinical outcome. Several studies indicate that AZM may accomplish its beneficial action in CF patients by impeding QS, thereby reducing the pathogenicity of P. aeruginosa. This led us to investigate whether QS inhibition is a common feature of antibiotics. We present the results of a screening of 12 antibiotics for their QS-inhibitory activities using a previously described QS inhibitor selector 1 strain. Three of the antibiotics tested, AZM, ceftazidime (CFT), and ciprofloxacin (CPR), were very active in the assay and were further examined for their effects on QS-regulated virulence factor production in P. aeruginosa. The effects of the three antibiotics administered at subinhibitory concentrations were investigated by use of DNA microarrays. Consistent results from the virulence factor assays, reverse transcription-PCR, and the DNA microarrays support the finding that AZM, CFT, and CPR decrease the expression of a range of QS-regulated virulence factors. The data suggest that the underlying mechanism may be mediated by changes in membrane permeability, thereby influencing the flux of N-3-oxo-dodecanoyl-L-homoserine lactone.
Figures

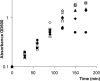
 ), 16 μg/ml AZM (▴), 0.04 μg/ml CPR (○), 0.08 μg/ml CPR (•), 0.25 μg/ml CFT (⋄), or 0.5 μg/ml CFT (⧫).
), 16 μg/ml AZM (▴), 0.04 μg/ml CPR (○), 0.08 μg/ml CPR (•), 0.25 μg/ml CFT (⋄), or 0.5 μg/ml CFT (⧫).
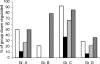
Similar articles
-
Impact of Azithromycin on the Quorum Sensing-Controlled Proteome of Pseudomonas aeruginosa.PLoS One. 2016 Jan 25;11(1):e0147698. doi: 10.1371/journal.pone.0147698. eCollection 2016. PLoS One. 2016. PMID: 26808156 Free PMC article.
-
Azithromycin blocks quorum sensing and alginate polymer formation and increases the sensitivity to serum and stationary-growth-phase killing of Pseudomonas aeruginosa and attenuates chronic P. aeruginosa lung infection in Cftr(-/-) mice.Antimicrob Agents Chemother. 2007 Oct;51(10):3677-87. doi: 10.1128/AAC.01011-06. Epub 2007 Jul 9. Antimicrob Agents Chemother. 2007. PMID: 17620382 Free PMC article.
-
Baicalin inhibits biofilm formation, attenuates the quorum sensing-controlled virulence and enhances Pseudomonas aeruginosa clearance in a mouse peritoneal implant infection model.PLoS One. 2017 Apr 28;12(4):e0176883. doi: 10.1371/journal.pone.0176883. eCollection 2017. PLoS One. 2017. PMID: 28453568 Free PMC article.
-
Central role of quorum sensing in regulating the production of pathogenicity factors in Pseudomonas aeruginosa.Future Microbiol. 2008 Feb;3(1):97-106. doi: 10.2217/17460913.3.1.97. Future Microbiol. 2008. PMID: 18230038 Review.
-
Revealing quorum-sensing networks in Pseudomonas aeruginosa infections through internal and external signals to prevent new resistance trends.Microbiol Res. 2024 Dec;289:127915. doi: 10.1016/j.micres.2024.127915. Epub 2024 Sep 24. Microbiol Res. 2024. PMID: 39342746 Review.
Cited by
-
Role of quorum sensing in bacterial infections.World J Clin Cases. 2015 Jul 16;3(7):575-98. doi: 10.12998/wjcc.v3.i7.575. World J Clin Cases. 2015. PMID: 26244150 Free PMC article. Review.
-
Cilostazol is a promising anti-pseudomonal virulence drug by disruption of quorum sensing.AMB Express. 2024 Aug 1;14(1):87. doi: 10.1186/s13568-024-01740-1. AMB Express. 2024. PMID: 39090255 Free PMC article.
-
Anti-inflammatory effects of antibacterials on human Bronchial epithelial cells.Respir Res. 2009 Sep 29;10(1):89. doi: 10.1186/1465-9921-10-89. Respir Res. 2009. PMID: 19788749 Free PMC article.
-
In vitro screens for quorum sensing inhibitors and in vivo confirmation of their effect.Nat Protoc. 2010 Feb;5(2):282-93. doi: 10.1038/nprot.2009.205. Epub 2010 Jan 28. Nat Protoc. 2010. PMID: 20134428
-
Design, green synthesis, and quorum sensing quenching potential of novel 2-oxo-pyridines containing a thiophene/furan scaffold and targeting a LasR gene on P. aeruginosa.RSC Adv. 2023 Sep 13;13(39):27363-27384. doi: 10.1039/d3ra04230h. eCollection 2023 Sep 8. RSC Adv. 2023. PMID: 37711372 Free PMC article.
References
-
- Andrade, S. S., R. N. Jones, A. C. Gales, and H. S. Sader. 2003. Increasing prevalence of antimicrobial resistance among Pseudomonas aeruginosa isolates in Latin American medical centres: 5 year report of the SENTRY Antimicrobial Surveillance Program (1997-2001). J. Antimicrob. Chemother. 52:140-141. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

