Reconfiguration of the pontomedullary respiratory network: a computational modeling study with coordinated in vivo experiments
- PMID: 18650310
- PMCID: PMC2576193
- DOI: 10.1152/jn.90416.2008
Reconfiguration of the pontomedullary respiratory network: a computational modeling study with coordinated in vivo experiments
Abstract
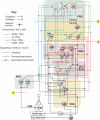
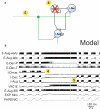
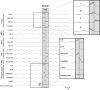
A large body of data suggests that the pontine respiratory group (PRG) is involved in respiratory phase-switching and the reconfiguration of the brain stem respiratory network. However, connectivity between the PRG and ventral respiratory column (VRC) in computational models has been largely ad hoc. We developed a network model with PRG-VRC connectivity inferred from coordinated in vivo experiments. Neurons were modeled in the "integrate-and-fire" style; some neurons had pacemaker properties derived from the model of Breen et al. We recapitulated earlier modeling results, including reproduction of activity profiles of different respiratory neurons and motor outputs, and their changes under different conditions (vagotomy, pontine lesions, etc.). The model also reproduced characteristic changes in neuronal and motor patterns observed in vivo during fictive cough and during hypoxia in non-rapid eye movement sleep. Our simulations suggested possible mechanisms for respiratory pattern reorganization during these behaviors. The model predicted that network- and pacemaker-generated rhythms could be co-expressed during the transition from gasping to eupnea, producing a combined "burst-ramp" pattern of phrenic discharges. To test this prediction, phrenic activity and multiple single neuron spike trains were monitored in vagotomized, decerebrate, immobilized, thoracotomized, and artificially ventilated cats during hypoxia and recovery. In most experiments, phrenic discharge patterns during recovery from hypoxia were similar to those predicted by the model. We conclude that under certain conditions, e.g., during recovery from severe brain hypoxia, components of a distributed network activity present during eupnea can be co-expressed with gasp patterns generated by a distinct, functionally "simplified" mechanism.
Figures








References
-
- Alheid GF, Gray PA, Jiang MC, Feldman JL, McCrimmon DR. Parvalbumin in respiratory neurons of the ventrolateral medulla of the adult rat. J Neurocytol 31: 693–717, 2002. - PubMed
-
- Alheid GF, Milsomb WK, McCrimmon DR. Pontine influences on breathing: an overview. Respir Physiol Neurobiol 143: 105–114, 2004. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

