Neuroinflammation and oxidation/nitration of alpha-synuclein linked to dopaminergic neurodegeneration
- PMID: 18650345
- PMCID: PMC2702093
- DOI: 10.1523/JNEUROSCI.0143-07.2008
Neuroinflammation and oxidation/nitration of alpha-synuclein linked to dopaminergic neurodegeneration
Abstract
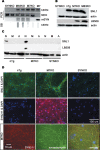
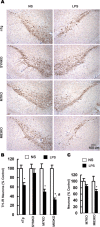
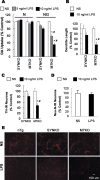
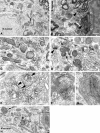
alpha-Synuclein (SYN) is the major component of Lewy bodies, the neuropathological hallmarks of Parkinson's disease (PD). Missense mutations and multiplications of the SYN gene cause autosomal dominant inherited PD. Thus, SYN is implicated in the pathogenesis of PD. However, the mechanism whereby SYN promotes neurodegeneration remains unclear. Familial PD with SYN gene mutations are rare because the majority of PD is sporadic and emerging evidence indicates that sporadic PD may result from genetic and environmental risk factors including neuroinflammation. Hence, we examined the relationship between SYN dysfunction and neuroinflammation in mediating dopaminergic neurodegeneration in mice and dopaminergic neuronal cultures derived from wild-type SYN and mutant A53T SYN transgenic mice in a murine SYN-null (SYNKO) background (M7KO and M83KO, respectively). Stereotaxic injection of an inflammagen, lipopolysaccharide, into substantia nigra of these SYN genetically engineered mice induced similar inflammatory reactions. In M7KO and M83KO, but not in SYNKO mice, the neuroinflammation was associated with dopaminergic neuronal death and the accumulation of insoluble aggregated SYN as cytoplasmic inclusions in nigral neurons. Nitrated/oxidized SYN was detected in these inclusions and abatement of microglia-derived nitric oxide and superoxide provided significant neuroprotection in neuron-glia cultures from M7KO mice. These data suggest that nitric oxide and superoxide released by activated microglia may be mediators that link inflammation and abnormal SYN in mechanisms of PD neurodegeneration. This study advances understanding of the role of neuroinflammation and abnormal SYN in the pathogenesis of PD and opens new avenues for the discovery of more effective therapies for PD.
Figures




Similar articles
-
Neuroinflammation and α-synuclein dysfunction potentiate each other, driving chronic progression of neurodegeneration in a mouse model of Parkinson's disease.Environ Health Perspect. 2011 Jun;119(6):807-14. doi: 10.1289/ehp.1003013. Epub 2011 Jan 18. Environ Health Perspect. 2011. PMID: 21245015 Free PMC article.
-
Cx3cr1-deficiency exacerbates alpha-synuclein-A53T induced neuroinflammation and neurodegeneration in a mouse model of Parkinson's disease.Glia. 2018 Aug;66(8):1752-1762. doi: 10.1002/glia.23338. Epub 2018 Apr 6. Glia. 2018. PMID: 29624735
-
Age-dependent neurodegeneration and neuroinflammation in a genetic A30P/A53T double-mutated α-synuclein mouse model of Parkinson's disease.Neurobiol Dis. 2022 Sep;171:105798. doi: 10.1016/j.nbd.2022.105798. Epub 2022 Jun 21. Neurobiol Dis. 2022. PMID: 35750147
-
Reprint of: revisiting oxidative stress and mitochondrial dysfunction in the pathogenesis of Parkinson disease-resemblance to the effect of amphetamine drugs of abuse.Free Radic Biol Med. 2013 Sep;62:186-201. doi: 10.1016/j.freeradbiomed.2013.05.042. Epub 2013 Jun 3. Free Radic Biol Med. 2013. PMID: 23743292 Review.
-
The contribution of alpha synuclein to neuronal survival and function - Implications for Parkinson's disease.J Neurochem. 2016 May;137(3):331-59. doi: 10.1111/jnc.13570. Epub 2016 Mar 23. J Neurochem. 2016. PMID: 26852372 Free PMC article. Review.
Cited by
-
α-Synuclein in Parkinson's disease.Cold Spring Harb Perspect Med. 2012 Feb;2(2):a009399. doi: 10.1101/cshperspect.a009399. Cold Spring Harb Perspect Med. 2012. PMID: 22355802 Free PMC article. Review.
-
Inflaming the diseased brain: a role for tainted melanins.Biochim Biophys Acta. 2015 May;1852(5):937-50. doi: 10.1016/j.bbadis.2015.01.004. Epub 2015 Jan 10. Biochim Biophys Acta. 2015. PMID: 25585261 Free PMC article.
-
A spirulina-enhanced diet provides neuroprotection in an α-synuclein model of Parkinson's disease.PLoS One. 2012;7(9):e45256. doi: 10.1371/journal.pone.0045256. Epub 2012 Sep 18. PLoS One. 2012. PMID: 23028885 Free PMC article.
-
Highly pathogenic H5N1 influenza virus can enter the central nervous system and induce neuroinflammation and neurodegeneration.Proc Natl Acad Sci U S A. 2009 Aug 18;106(33):14063-8. doi: 10.1073/pnas.0900096106. Epub 2009 Aug 10. Proc Natl Acad Sci U S A. 2009. PMID: 19667183 Free PMC article.
-
Microglia affect α-synuclein cell-to-cell transfer in a mouse model of Parkinson's disease.Mol Neurodegener. 2019 Aug 16;14(1):34. doi: 10.1186/s13024-019-0335-3. Mol Neurodegener. 2019. PMID: 31419995 Free PMC article.
References
-
- Abeliovich A, Schmitz Y, Fariñas I, Choi-Lundberg D, Ho WH, Castillo PE, Shinsky N, Verdugo JM, Armanini M, Ryan A, Hynes M, Phillips H, Sulzer D, Rosenthal A. Mice lacking alpha-synuclein display functional deficits in the nigrostriatal dopamine system. Neuron. 2000;25:239–252. - PubMed
-
- Arawaka S, Wada M, Goto S, Karube H, Sakamoto M, Ren CH, Koyama S, Nagasawa H, Kimura H, Kawanami T, Kurita K, Tajima K, Daimon M, Baba M, Kido T, Saino S, Goto K, Asao H, Kitanaka C, Takashita E, et al. The role of G-protein-coupled receptor kinase 5 in pathogenesis of sporadic Parkinson's disease. J Neurosci. 2006;26:9227–9238. - PMC - PubMed
-
- Block ML, Wu X, Pei Z, Li G, Wang T, Qin L, Wilson B, Yang J, Hong JS, Veronesi B. Nanometer size diesel exhaust particles are selectively toxic to dopaminergic neurons: the role of microglia, phagocytosis, and NADPH oxidase. FASEB J. 2004;18:1618–1620. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous
