Characterization of side population cells from human airway epithelium
- PMID: 18653771
- PMCID: PMC2849005
- DOI: 10.1634/stemcells.2008-0171
Characterization of side population cells from human airway epithelium
Abstract
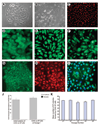
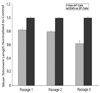
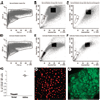
The airway epithelium is the first line of contact with the inhaled external environment and is continuously exposed to and injured by pollutants, allergens, and viruses. However, little is known about epithelial repair and in particular the identity and role of tissue resident stem/progenitor cells that may contribute to epithelial regeneration. The aims of the present study were to identify, isolate, and characterize side population (SP) cells in human tracheobronchial epithelium. Epithelial cells were obtained from seven nontransplantable healthy lungs and four asthmatic lungs by pronase digestion. SP cells were identified by verapamil-sensitive efflux of the DNA-binding dye Hoechst 33342. Using flow cytometry, CD45(-) SP, CD45(+) SP, and non-SP cells were isolated and sorted. CD45(-) SP cells made up 0.12% +/- 0.01% of the total epithelial cell population in normal airway but 4.1% +/- 0.06% of the epithelium in asthmatic airways. All CD45(-) SP cells showed positive staining for epithelial-specific markers cytokeratin-5, E-cadherin, ZO-1, and p63. CD45(-) SP cells exhibited stable telomere length and increased colony-forming and proliferative potential, undergoing population expansion for at least 16 consecutive passages. In contrast with non-SP cells, fewer than 100 CD45(-) SP cells were able to generate a multilayered and differentiated epithelium in air-liquid interface culture. SP cells are present in human tracheobronchial epithelium, exhibit both short- and long-term proliferative potential, and are capable of generation of differentiated epithelium in vitro. The number of SP cells is significantly greater in asthmatic airways, providing evidence of dysregulated resident SP cells in the asthmatic epithelium. Disclosure of potential conflicts of interest is found at the end of this article.
Conflict of interest statement
The authors indicate no potential conflicts of interest.
Figures


References
-
- Knight DA, Holgate ST. The airway epithelium: Structural and functional properties in health and disease. Respirology. 2003;8:432–446. - PubMed
-
- Hackett TL, Knight DA. The role of epithelial injury and repair in the origins of asthma. Curr Opin Allergy Clin Immunol. 2007;7:63–68. - PubMed
-
- Barbato A, Turato G, Baraldo S, et al. Epithelial damage and angiogenesis in the airways of children with asthma. Am J Respir Crit Care Med. 2006;174:975–981. - PubMed
-
- Lipkin M. Growth and development of gastrointestinal cells. Annu Rev Physiol. 1985;47:175–197. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

