Chlamydiae and polymorphonuclear leukocytes: unlikely allies in the spread of chlamydial infection
- PMID: 18657107
- PMCID: PMC2925246
- DOI: 10.1111/j.1574-695X.2008.00459.x
Chlamydiae and polymorphonuclear leukocytes: unlikely allies in the spread of chlamydial infection
Abstract
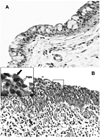
While much is known about the attachment of the chlamydiae to the host cell and intracellular events during the developmental cycle, little is known about the mechanism(s) by which elementary bodies exit the cell. In this report, we use the guinea-pig conjunctival model of Chlamydia caviae infection to present in vivo ultrastructural evidence supporting two mechanisms for release of chlamydiae from the mucosal epithelia. Four days after infection, histopathologic observation shows an intense infiltration of polymorphonuclear leukocytes (PMN) in the conjunctival epithelium. Using transmission electron microscopy, a gradient-directed PMN response to chlamydiae-infected epithelial cells was observed. As PMN infiltration intensifies, epithelial hemidesmosome/integrin/focal adhesion adherence with the basal lamina is disconnected and PMNs literally lift off and release infected superficial epithelia from the mucosa. Many of these infected cells appear to be healthy with intact microvilli, nuclei, and mitochondria. While lysis of some infected cells occurs with release of chlamydiae into the extracellular surface milieu, the majority of infected cells are pushed off the epithelium. We propose that PMNs play an active role in detaching infected cells from the epithelium and that these infected cells eventually die releasing organisms but, in the process, move to new tissue sites via fluid dynamics.
Figures







References
-
- Abu El-Asrar AM, Geboes K, al Kharashi SA, Tabbara KF, Missotten L. Collagen content and types in trachomatous conjunctivitis. Eye. 1998;12(Pt 4):735–739. - PubMed
-
- Barnhart KT, Stolpen A, Pretorius ES, Malamud D. Distribution of a spermicide containing Nonoxynol-9 in the vaginal canal and the upper female reproductive tract. Hum.Reprod. 2001;16:1151–1154. - PubMed
-
- Barron AL, White HJ, Rank RG, Soloff BL. Target tissues associated with genital infection of female guinea pigs by the chlamydial agent of guinea pig inclusion conjunctivitis. J.Infect.Dis. 1979;139:60–68. - PubMed
-
- Buchholz KR, Stephens RS. Activation of the host cell proinflammatory interleukin-8 response by Chlamydia trachomatis. Cell Microbiol. 2006;8:1768–1779. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
