Intrinsic ruminal innervation in ruminants of different feeding types
- PMID: 18657258
- PMCID: PMC2644769
- DOI: 10.1111/j.1469-7580.2008.00959.x
Intrinsic ruminal innervation in ruminants of different feeding types
Abstract
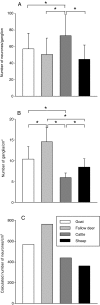
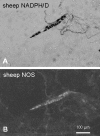
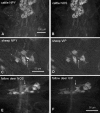
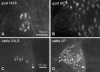
According to their feeding habits, ruminants can be classified as grazers, concentrate selectors and those of intermediate type. The different feeding types are reflected in distinct anatomical properties of the forestomachs. The present study was designed to investigate whether the intrinsic innervation patterns of the rumen (the main part of the forestomach) differ between intermediate types and grazers. Myenteric plexus preparations from the rumen of goats (intermediate type), fallow deer (intermediate type), cattle (grazer) and sheep (grazer) were analysed by immunohistochemical detection of the following antigens: Hu-protein (HuC/D), choline acetyltransferase (ChAT), nitric oxide synthase (NOS), vasoactive intestinal peptide (VIP), neuropeptide Y (NPY), substance P (SP), calbindin (CALB) and somatostatin (SOM). Myenteric ganglia of cattle contained 73 +/- 6 neurons per ganglion, whereas the ganglia of sheep were significantly smaller (45 +/- 18 neurons per ganglion). The ganglion density of the myenteric plexus was highest in fallow deer (15 +/- 3 ganglia per cm(2)) and lowest in cattle (6 +/- 1 ganglia per cm(2)). All myenteric neurons were either ChAT or NOS positive. The proportion of NOS-positive neurons was significantly lower in sheep (29.5 +/- 8.2% of all neurons) than in goats (44.2 +/- 9.8%). In all species, additional analysis of the different neuropeptides revealed the following subpopulations in descending order of percentile appearance: ChAT/SP > NOS/VIP/NPY > ChAT/- > NOS/NPY. Expression of CALB was detected in a minority of the ChAT-positive neurons in all species. Somatostatin immunoreactive somata were found only in preparations obtained from fallow deer and sheep. These data suggest that the rumen of grazers is under stronger cholinergic control than the rumen of species belonging to the intermediate type, although most subpopulations of neurons are present in all species. However, whether the strong mixing patterns of low quality roughage during digestion are enabled by the prominent excitatory input of the rumen of grazers requires elucidation in further studies.
Figures



Similar articles
-
Calbindin-immunoreactive neurones in the ovine rumen.Anat Rec A Discov Mol Cell Evol Biol. 2004 Jun;278(2):528-32. doi: 10.1002/ar.a.20048. Anat Rec A Discov Mol Cell Evol Biol. 2004. PMID: 15164340
-
Age-associated plasticity in the intrinsic innervation of the ovine rumen.J Anat. 2003 Sep;203(3):277-82. doi: 10.1046/j.1469-7580.2003.00218.x. J Anat. 2003. PMID: 14529045 Free PMC article.
-
Ruminal muscle of sheep is innervated by non-polarized pathways of cholinergic and nitrergic myenteric neurones.Cell Tissue Res. 2002 Sep;309(3):347-54. doi: 10.1007/s00441-002-0554-7. Epub 2002 Jul 13. Cell Tissue Res. 2002. PMID: 12195290
-
Mucosa of the guinea pig gastric corpus is innervated by myenteric neurones with specific neurochemical coding and projection preferences.J Comp Neurol. 1999 Aug 2;410(3):489-502. doi: 10.1002/(sici)1096-9861(19990802)410:3<489::aid-cne10>3.0.co;2-s. J Comp Neurol. 1999. PMID: 10404414
-
Reticular groove and reticulum are innervated by myenteric neurons with different neurochemical codes.Anat Rec A Discov Mol Cell Evol Biol. 2003 Oct;274(2):917-22. doi: 10.1002/ar.a.10104. Anat Rec A Discov Mol Cell Evol Biol. 2003. PMID: 12973715
Cited by
-
Immunohistochemical evaluation of the goat forestomach during prenatal development.J Vet Sci. 2014;15(1):35-43. doi: 10.4142/jvs.2014.15.1.35. Epub 2013 Oct 18. J Vet Sci. 2014. PMID: 24136206 Free PMC article.
References
-
- Brehmer A, Croner R, Dimmler A, Papadopoulos T, Schrodl F, Neuhuber W. Immunohistochemical characterization of putative primary afferent (sensory) myenteric neurons in human small intestine. Auton Neurosci. 2004;112:49–59. - PubMed
-
- Brookes SJ. Classes of enteric nerve cells in the guinea-pig small intestine. Anat Rec. 2001;262:58–70. - PubMed
-
- Brookes SJ, Hennig G, Schemann M. Identification of motor neurons to the circular muscle of the guinea pig gastric corpus. J Comp Neurol. 1998;397:268–280. - PubMed
-
- Denac M, Marti J, Scharrer E. Relaxation of ruminal smooth muscle by vasoactive intestinal polypeptide (VIP) Zentralbl Veterinarmed A. 1987;34:317–320. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

