Transglutaminases and transglutaminase-catalyzed cross-links colocalize with the pathological lesions in Alzheimer's disease brain
- PMID: 18673368
- PMCID: PMC8094859
- DOI: 10.1111/j.1750-3639.2008.00197.x
Transglutaminases and transglutaminase-catalyzed cross-links colocalize with the pathological lesions in Alzheimer's disease brain
Abstract
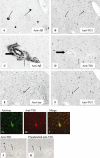
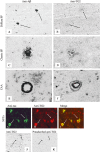
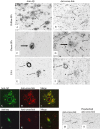
Alzheimer's disease (AD) is characterized by pathological lesions, in particular senile plaques (SPs), cerebral amyloid angiopathy (CAA) and neurofibrillary tangles (NFTs), predominantly consisting of self-aggregated proteins amyloid beta (Abeta) and tau, respectively. Transglutaminases (TGs) are inducible enzymes, capable of modifying conformational and/or structural properties of proteins by inducing molecular covalent cross-links. Both Abeta and tau are substrates for TG cross-linking activity, which links TGs to the aggregation process of both proteins in AD brain. The aim of this study was to investigate the association of transglutaminase 1 (TG1), transglutaminase 2 (TG2) and TG-catalyzed cross-links with the pathological lesions of AD using immunohistochemistry. We observed immunoreactivity for TG1, TG2 and TG-catalyzed cross-links in NFTs. In addition, both TG2 and TG-catalyzed cross-links colocalized with Abeta in SPs. Furthermore, both TG2 and TG-catalyzed cross-links were associated with CAA. We conclude that these TGs demonstrate cross-linking activity in AD lesions, which suggests that both TG1 and TG2 are likely involved in the protein aggregation processes underlying the formation of SPs, CAA and/or NFTs in AD brain.
Figures

Similar articles
-
Tissue transglutaminase-catalysed cross-linking induces Apolipoprotein E multimers inhibiting Apolipoprotein E's protective effects towards amyloid-beta-induced toxicity.J Neurochem. 2015 Sep;134(6):1116-28. doi: 10.1111/jnc.13203. Epub 2015 Jul 7. J Neurochem. 2015. PMID: 26088696
-
Transglutaminase 1 and its regulator tazarotene-induced gene 3 localize to neuronal tau inclusions in tauopathies.J Pathol. 2012 Jan;226(1):132-42. doi: 10.1002/path.2984. Epub 2011 Oct 18. J Pathol. 2012. PMID: 22009441 Free PMC article.
-
Binding of curcumin to senile plaques and cerebral amyloid angiopathy in the aged brain of various animals and to neurofibrillary tangles in Alzheimer's brain.J Vet Med Sci. 2012 Jan;74(1):51-7. doi: 10.1292/jvms.11-0307. Epub 2011 Sep 2. J Vet Med Sci. 2012. PMID: 21891973
-
Alzheimer's Disease Neuropathological Change in Aged Non-Primate Mammals.Int J Mol Sci. 2024 Jul 25;25(15):8118. doi: 10.3390/ijms25158118. Int J Mol Sci. 2024. PMID: 39125687 Free PMC article. Review.
-
Review on Alzheimer's disease: Inhibition of amyloid beta and tau tangle formation.Int J Biol Macromol. 2021 Jan 15;167:382-394. doi: 10.1016/j.ijbiomac.2020.11.192. Epub 2020 Dec 2. Int J Biol Macromol. 2021. PMID: 33278431 Review.
Cited by
-
Possible role of the transglutaminases in the pathogenesis of Alzheimer's disease and other neurodegenerative diseases.Int J Alzheimers Dis. 2011 Feb 16;2011:865432. doi: 10.4061/2011/865432. Int J Alzheimers Dis. 2011. PMID: 21350675 Free PMC article.
-
Aberrant calcium signaling by transglutaminase-mediated posttranslational modification of inositol 1,4,5-trisphosphate receptors.Proc Natl Acad Sci U S A. 2014 Sep 23;111(38):E3966-75. doi: 10.1073/pnas.1409730111. Epub 2014 Sep 8. Proc Natl Acad Sci U S A. 2014. PMID: 25201980 Free PMC article.
-
Physiological, pathological, and structural implications of non-enzymatic protein-protein interactions of the multifunctional human transglutaminase 2.Cell Mol Life Sci. 2015 Aug;72(16):3009-35. doi: 10.1007/s00018-015-1909-z. Epub 2015 May 6. Cell Mol Life Sci. 2015. PMID: 25943306 Free PMC article. Review.
-
Discovery and characterization of a photo-oxidative histidine-histidine cross-link in IgG1 antibody utilizing ¹⁸O-labeling and mass spectrometry.Anal Chem. 2014 May 20;86(10):4940-8. doi: 10.1021/ac500334k. Epub 2014 May 2. Anal Chem. 2014. PMID: 24738698 Free PMC article.
-
Tissue Transglutaminase and Its Product Isopeptide Are Increased in Alzheimer's Disease and APPswe/PS1dE9 Double Transgenic Mice Brains.Mol Neurobiol. 2016 Oct;53(8):5066-78. doi: 10.1007/s12035-015-9413-x. Epub 2015 Sep 19. Mol Neurobiol. 2016. PMID: 26386840 Free PMC article.
References
-
- Aeschlimann D, Thomazy V (2000) Protein crosslinking in assembly and remodelling of extracellular matrices: the role of transglutaminases. Connect Tissue Res 41:1–27. - PubMed
-
- Akimov SS, Belkin AM (2001) Cell surface tissue transglutaminase is involved in adhesion and migration of monocytic cells on fibronectin. Blood 98:1567–1576. - PubMed
-
- Atwood CS, Moir RD, Huang X, Scarpa RC, Bacarra NM, Romano DM et al (1998) Dramatic aggregation of Alzheimer abeta by Cu(II) is induced by conditions representing physiological acidosis. J Biol Chem 273:12817–12826. - PubMed
-
- Baumgartner W, Weth A (2007) Transglutaminase 1 stabilizes beta‐actin in endothelial cells correlating with a stabilization of intercellular junctions. J Vasc Res 3:234–240. - PubMed
-
- Bonelli RM, Aschoff A, Niederwieser G, Heuberger C, Jirikowski G (2002) Cerebrospinal fluid tissue transglutaminase as a biochemical marker for Alzheimer's disease. Neurobiol Dis 11:106–110. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

