In vitro polydeoxyribonucleotide effects on human pre-adipocytes
- PMID: 18673371
- PMCID: PMC6496018
- DOI: 10.1111/j.1365-2184.2008.00547.x
In vitro polydeoxyribonucleotide effects on human pre-adipocytes
Abstract
Objectives: Adipose tissue is the most abundant and accessible source of adult stem cells. Human processed lipoaspirate contains pre-adipocytes that possess one of the a characteristic pathways of multipotent adult stem cells and are able to differentiate in vitro into mesenchymal and also neurogenic lineages. Because stem cells have great potential for use in tissue repair and regeneration, it would be significant to be able to obtain large amounts of these cells in vitro. As demonstrated previously, purine nucleosides and nucleotides mixtures can act as mitogens for several cell types. The aim of this study was to evaluate the effects of polydeoxyribonucleotides (PDRN), at appropriate concentrations, on human pre-adipocytes grown in a controlled medium, also using different passages, so as to investigate the relationship between the effect of this compound and cellular senescence, which is the phenomenon when normal diploid cells lose the ability to divide further.
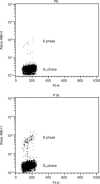
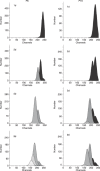
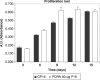
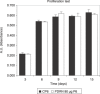
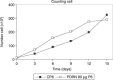
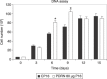
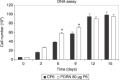
Materials and methods: Human pre-adipocytes were obtained by liposuction. Cells from different culture passages (P6 and P16) were treated with PDRN at different experimental times. Cell number was evaluated for each sample by direct counting after trypan blue treatment. DNA assay and the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide test were also carried out in all cases.
Results and conclusions: PDRN seemed to promote proliferation of human pre-adipocytes at both passages, but cell population growth increased in pre-adipocyte at P16, after 9 days as compared to control. Our data suggest that PDRN could act as a pre-adipocyte growth stimulator.
Figures



References
-
- Ashjian PH, Elbarbary AS, Edmonds B, Deugarte D, Zhu M, Zuk PA, Lorenz HP, Benhaim P, Hedrick MH (2003) In vitro differentiation of human processed lipoaspirate cells into early neural progenitors. Plast. Reconstr. Surg. 111, 1922–1931. - PubMed
-
- Astori G, Vignati F, Bardelli S, Tubio M, Gola M, Albertini V, Bambi F, Scali G, Castelli D, Rasini V, Soldati G, Moccetti T (2007) ‘In vitro’ and multicolor phenotypic characterization of cell subpopulations identified in fresh human adipose tissue stromal vascular fraction and in the derived mesenchymal stem cells. J. Transl. Med. 5, 55. - PMC - PubMed
-
- Belletti S, Uggeri J, Gatti R, Govoni P, Guizzardi S (2007) Polydeoxyribonucleotide promotes cyclobutane pyrimidine dimer repair in UVB‐exposed dermal fibroblasts. Photodermatol. Photoimmunol. Photomed. 23, 242–249. - PubMed
-
- Bigliardi P (1982) Treatment of acute radiodermatitis of first and second degrees with semi‐greasy placenta ointment. Int. J. Tissue React. 4, 153–154. - PubMed
-
- Bitto A, Minatoli L, Polito F, Altavilla D, Cattarini G, Squadrito F (2006) Polydeoxyribonucleotide improve angiogenesis and wound healing in experimental burn wounds In: 8th International Symposium on Adenosine and Adenine Nucleotides. Ferrara, Italy: Springer.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

