Nonspecific interaction of prefibrillar amyloid aggregates with glutamatergic receptors results in Ca2+ increase in primary neuronal cells
- PMID: 18676369
- PMCID: PMC2662062
- DOI: 10.1074/jbc.M803992200
Nonspecific interaction of prefibrillar amyloid aggregates with glutamatergic receptors results in Ca2+ increase in primary neuronal cells
Abstract
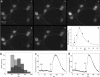
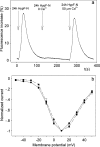
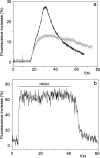
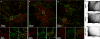
It is widely reported that the Ca(2+) increase following nonspecific cell membrane permeabilization is among the earliest biochemical modifications in cells exposed to toxic amyloid aggregates. However, more recently receptors with Ca(2+) channel activity such as alpha-amino-3-hydroxy-5-methylisoxazole-4-propionic acid (AMPA), N-methyl D-aspartate (NMDA), ryanodine, and inositol 1,4,5-trisphosphate receptors have been proposed as mediators of the Ca(2+) increase in neuronal cells challenged with beta-amyloid peptides. We previously showed that prefibrillar aggregates of proteins not associated with amyloid diseases are toxic to exposed cells similarly to comparable aggregates of disease-associated proteins. In particular, prefibrillar aggregates of the prokaryotic HypF-N were shown to be toxic to different cultured cell lines by eliciting Ca(2+) and reactive oxygen species increases. This study was aimed at assessing whether NMDA and AMPA receptor activations could be considered a generic feature of cell interaction with amyloid aggregates rather than a specific effect of some aggregated protein. Therefore, we investigated whether NMDA and AMPA receptors were involved in the Ca(2+) increase following exposure of rat cerebellar granule cells to HypF-N prefibrillar aggregates. We found that the intracellular Ca(2+) increase was associated with the early activation of NMDA and AMPA receptors, although some nonspecific membrane permeabilization was also observed at longer times of exposure. This result matched a significant co-localization of the aggregates with both receptors on the plasma membrane. Our data support the possibility that glutamatergic channels are generic sites of interaction with the cell membrane of prefibrillar aggregates of different peptides and proteins as well as the key structures responsible for the resulting early membrane permeabilization to Ca(2+).
Figures
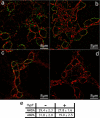
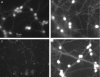

Similar articles
-
Different ataxin-3 amyloid aggregates induce intracellular Ca(2+) deregulation by different mechanisms in cerebellar granule cells.Biochim Biophys Acta. 2013 Dec;1833(12):3155-3165. doi: 10.1016/j.bbamcr.2013.08.019. Epub 2013 Sep 11. Biochim Biophys Acta. 2013. PMID: 24035922
-
Calcium entry through a subpopulation of AMPA receptors desensitized neighbouring NMDA receptors in rat dorsal horn neurons.J Physiol. 1995 Jun 1;485 ( Pt 2)(Pt 2):373-81. doi: 10.1113/jphysiol.1995.sp020736. J Physiol. 1995. PMID: 7545229 Free PMC article.
-
Overactivation of alpha-amino-3-hydroxy-5-methylisoxazole-4-propionate and N-methyl-D-aspartate but not kainate receptors inhibits phosphatidylcholine synthesis before excitotoxic neuronal death.J Neurochem. 2001 Apr;77(1):13-22. doi: 10.1046/j.1471-4159.2001.t01-2-00187.x. J Neurochem. 2001. PMID: 11279257
-
Soluble Oligomers Require a Ganglioside to Trigger Neuronal Calcium Overload.J Alzheimers Dis. 2017;60(3):923-938. doi: 10.3233/JAD-170340. J Alzheimers Dis. 2017. PMID: 28922156
-
Prefibrillar amyloid protein aggregates share common features of cytotoxicity.J Biol Chem. 2004 Jul 23;279(30):31374-82. doi: 10.1074/jbc.M400348200. Epub 2004 May 6. J Biol Chem. 2004. PMID: 15133040
Cited by
-
β-Amyloid (1-40) peptide interactions with supported phospholipid membranes: a single-molecule study.Biophys J. 2012 Oct 3;103(7):1500-9. doi: 10.1016/j.bpj.2012.08.051. Epub 2012 Oct 2. Biophys J. 2012. PMID: 23062342 Free PMC article.
-
Amyloid oligomer neurotoxicity, calcium dysregulation, and lipid rafts.Int J Alzheimers Dis. 2011 Feb 8;2011:906964. doi: 10.4061/2011/906964. Int J Alzheimers Dis. 2011. PMID: 21331330 Free PMC article.
-
Different Molecular Mechanisms Mediate Direct or Glia-Dependent Prion Protein Fragment 90-231 Neurotoxic Effects in Cerebellar Granule Neurons.Neurotox Res. 2017 Oct;32(3):381-397. doi: 10.1007/s12640-017-9749-2. Epub 2017 May 25. Neurotox Res. 2017. PMID: 28540665
-
Protein folding and misfolding on surfaces.Int J Mol Sci. 2008 Dec;9(12):2515-2542. doi: 10.3390/ijms9122515. Epub 2008 Dec 9. Int J Mol Sci. 2008. PMID: 19330090 Free PMC article.
-
Excitotoxicity through NMDA receptors mediates cerebellar granule neuron apoptosis induced by prion protein 90-231 fragment.Neurotox Res. 2013 May;23(4):301-14. doi: 10.1007/s12640-012-9340-9. Epub 2012 Aug 2. Neurotox Res. 2013. PMID: 22855343
References
-
- Stefani, M., and Dobson, C. M. (2003) J. Mol. Med. 81 678-699 - PubMed
-
- Stefani, M. (2004) Biochim. Biophys. Acta 1739 5-25 - PubMed
-
- Walsh, D. M., and Selkoe, D. J. (2004) Protein Pept. Lett. 11 213-228 - PubMed
-
- Kayed, R., Head, E., Thompson, J. L., McIntire, T. M., Milton, S. C., Cotman, C. W., and Glabe, C. G. (2003) Science 300 486-489 - PubMed
-
- Giovannini, M. G., Scali, C., Prosperi, C., Bellucci, A., Vannucchi, M. G., Rosi, S., Pepeu, G., and Casamenti, F. (2002) Neurobiol. Dis. 11 257-274 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

