Novelties of the flowering plant pollen tube underlie diversification of a key life history stage
- PMID: 18678915
- PMCID: PMC2516219
- DOI: 10.1073/pnas.0800036105
Novelties of the flowering plant pollen tube underlie diversification of a key life history stage
Abstract
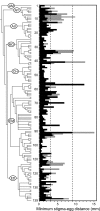
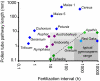
The origin and rapid diversification of flowering plants has puzzled evolutionary biologists, dating back to Charles Darwin. Since that time a number of key life history and morphological traits have been proposed as developmental correlates of the extraordinary diversity and ecological success of angiosperms. Here, I identify several innovations that were fundamental to the evolutionary lability of angiosperm reproduction, and hence to their diversification. In gymnosperms pollen reception must be near the egg largely because sperm swim or are transported by pollen tubes that grow at very slow rates (< approximately 20 microm/h). In contrast, pollen tube growth rates of taxa in ancient angiosperm lineages (Amborella, Nuphar, and Austrobaileya) range from approximately 80 to 600 microm/h. Comparative analyses point to accelerated pollen tube growth rate as a critical innovation that preceded the origin of the true closed carpel, long styles, multiseeded ovaries, and, in monocots and eudicots, much faster pollen tube growth rates. Ancient angiosperm pollen tubes all have callosic walls and callose plugs (in contrast, no gymnosperms have these features). The early association of the callose-walled growth pattern with accelerated pollen tube growth rate underlies a striking repeated pattern of faster and longer-distance pollen tube growth often within solid pathways in phylogenetically derived angiosperms. Pollen tube innovations are a key component of the spectacular diversification of carpel (flower and fruit) form and reproductive cycles in flowering plants.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Stebbins GL. Flowering Plants: Evolution above the Species Level. Cambridge, MA: Harvard Univ Press; 1974.
-
- Takhtajan A. In: Origin and Early Evolution of Angiosperms. Beck CB, editor. New York: Columbia Univ Press; 1976. pp. 207–219.
-
- Doyle JA. Origin of angiosperms. Ann Rev Ecol Syst. 1978;9:365–392.
-
- Favre-Ducharte M. Time relations and sexual reproduction in Cichorium and other angiosperms as compared with Archegoniates. Phytomorphology. 1979;29:166–178.
-
- Haig D, Westoby M. Selective forces in the emergence of the seed habit. Biol J Linn Soc. 1989;38:215–238.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

