Development and tissue origins of the mammalian cranial base
- PMID: 18680740
- PMCID: PMC2847450
- DOI: 10.1016/j.ydbio.2008.07.016
Development and tissue origins of the mammalian cranial base
Abstract
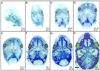
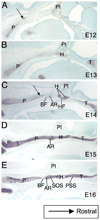
The vertebrate cranial base is a complex structure composed of bone, cartilage and other connective tissues underlying the brain; it is intimately connected with development of the face and cranial vault. Despite its central importance in craniofacial development, morphogenesis and tissue origins of the cranial base have not been studied in detail in the mouse, an important model organism. We describe here the location and time of appearance of the cartilages of the chondrocranium. We also examine the tissue origins of the mouse cranial base using a neural crest cell lineage cell marker, Wnt1-Cre/R26R, and a mesoderm lineage cell marker, Mesp1-Cre/R26R. The chondrocranium develops between E11 and E16 in the mouse, beginning with development of the caudal (occipital) chondrocranium, followed by chondrogenesis rostrally to form the nasal capsule, and finally fusion of these two parts via the midline central stem and the lateral struts of the vault cartilages. X-Gal staining of transgenic mice from E8.0 to 10 days post-natal showed that neural crest cells contribute to all of the cartilages that form the ethmoid, presphenoid, and basisphenoid bones with the exception of the hypochiasmatic cartilages. The basioccipital bone and non-squamous parts of the temporal bones are mesoderm derived. Therefore the prechordal head is mostly composed of neural crest-derived tissues, as predicted by the New Head Hypothesis. However, the anterior location of the mesoderm-derived hypochiasmatic cartilages, which are closely linked with the extra-ocular muscles, suggests that some tissues associated with the visual apparatus may have evolved independently of the rest of the "New Head".
Figures







References
-
- Abzhanov A, Tabin CJ. Shh and Fgf8 act synergistically to drive cartilage outgrowth during cranial development. Devel. Biol. 2004;273(1):134–148. - PubMed
-
- Abzhanov A, Cordero DR, Sen J, Tabin CJ, Helms JA. Cross-regulatory interactions between Fgf8 and Shh in the avian frontonasal prominence. Congenit.Anom. 2007;47(4):136–148. - PubMed
-
- Barteczko K, Jacob M. Comparative study of shape, course, and disintegration of the rostral notochord in some vertebrates, especially humans. Anat. Embryol. 1999;200:345–366. - PubMed
-
- Belo JA, Leyns L, Yamada G, De Robertis EM. The prechordal midline of the chondrocranium is defective in Goosecoid-1 mouse mutants. Mech Dev. 1998;71(1–2):15–25. - PubMed
-
- Bohme K, Li Y, Oh PS, Olsen BR. Primary structure of the long and short splice variants of mouse collagen XII and their tissue-specific expression during embryonic development. Dev Dyn. 1995;204(4):432–445. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

