Synergistic effects of Pten loss and WNT/CTNNB1 signaling pathway activation in ovarian granulosa cell tumor development and progression
- PMID: 18687666
- PMCID: PMC2577137
- DOI: 10.1093/carcin/bgn186
Synergistic effects of Pten loss and WNT/CTNNB1 signaling pathway activation in ovarian granulosa cell tumor development and progression
Abstract
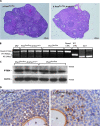
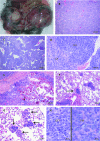
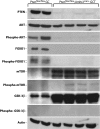
The mechanisms of granulosa cell tumor (GCT) development may involve the dysregulation of signaling pathways downstream of follicle-stimulating hormone, including the phosphoinosite-3 kinase (PI3K)/AKT pathway. To test this hypothesis, a genetically engineered mouse model was created to derepress the PI3K/AKT pathway in granulosa cells by conditional targeting of the PI3K antagonist gene Pten (Pten(flox/flox);Amhr2(cre/+)). The majority of Pten(flox/flox);Amhr2(cre/+) mice featured no ovarian anomalies, but occasionally ( approximately 7%) developed aggressive, anaplastic GCT with pulmonary metastases. The expression of the PI3K/AKT downstream effector FOXO1 was abrogated in Pten(flox/flox);Amhr2(cre/+) GCT, indicating a mechanism by which GCT cells may increase proliferation and evade apoptosis. To relate these findings to spontaneously occurring GCT, analyses of PTEN and phospho-AKT expression were performed on human and equine tumors. Although PTEN loss was not detected, many GCT (2/5 human, 7/17 equine) featured abnormal nuclear or perinuclear localization of phospho-AKT, suggestive of altered PI3K/AKT activity. As inappropriate activation of WNT/CTNNB1 signaling causes late-onset GCT development and cross talk between the PI3K/AKT and WNT/CTNNB1 pathways has been reported, we tested whether these pathways could synergize in GCT. Activation of both the PI3K/AKT and WNT/CTNNB1 pathways in the granulosa cells of a mouse model (Pten(flox/flox);Ctnnb1(flox(ex3)/+);Amhr2(cre/+)) resulted in the development of GCT similar to those observed in Pten(flox/flox);Amhr2(cre/+) mice, but with 100% penetrance, perinatal onset, extremely rapid growth and the ability to spread by seeding into the abdominal cavity. These data indicate a synergistic effect of dysregulated PI3K/AKT and WNT/CTNNB1 signaling in the development and progression of GCT and provide the first animal models for metastatic GCT.
Figures

Similar articles
-
Dominant-stable beta-catenin expression causes cell fate alterations and Wnt signaling antagonist expression in a murine granulosa cell tumor model.Cancer Res. 2006 Feb 15;66(4):1964-73. doi: 10.1158/0008-5472.CAN-05-3493. Cancer Res. 2006. PMID: 16488995
-
Dysregulation of WNT/CTNNB1 and PI3K/AKT signaling in testicular stromal cells causes granulosa cell tumor of the testis.Carcinogenesis. 2009 May;30(5):869-78. doi: 10.1093/carcin/bgp051. Epub 2009 Feb 23. Carcinogenesis. 2009. PMID: 19237610 Free PMC article.
-
Misregulated Wnt/beta-catenin signaling leads to ovarian granulosa cell tumor development.Cancer Res. 2005 Oct 15;65(20):9206-15. doi: 10.1158/0008-5472.CAN-05-1024. Cancer Res. 2005. PMID: 16230381
-
WNT signaling in ovarian follicle biology and tumorigenesis.Trends Endocrinol Metab. 2010 Jan;21(1):25-32. doi: 10.1016/j.tem.2009.08.005. Epub 2009 Oct 28. Trends Endocrinol Metab. 2010. PMID: 19875303 Review.
-
The molecular mechanism of ovarian granulosa cell tumors.J Ovarian Res. 2018 Feb 6;11(1):13. doi: 10.1186/s13048-018-0384-1. J Ovarian Res. 2018. PMID: 29409506 Free PMC article. Review.
Cited by
-
Combined comparative genomic hybridization and transcriptomic analyses of ovarian granulosa cell tumors point to novel candidate driver genes.BMC Cancer. 2015 Apr 10;15:251. doi: 10.1186/s12885-015-1283-0. BMC Cancer. 2015. PMID: 25884336 Free PMC article.
-
Complex immunohistochemical and molecular study on 5 cases of ovarian juvenile granulosa cell tumors reveals a consistent alteration in the PI3K/AKT/mTOR signaling pathway.Diagn Pathol. 2025 Jan 8;20(1):3. doi: 10.1186/s13000-025-01599-1. Diagn Pathol. 2025. PMID: 39773640 Free PMC article.
-
Decidual PTEN expression is required for trophoblast invasion in the mouse.Am J Physiol Endocrinol Metab. 2010 Dec;299(6):E936-46. doi: 10.1152/ajpendo.00255.2010. Epub 2010 Sep 21. Am J Physiol Endocrinol Metab. 2010. PMID: 20858757 Free PMC article.
-
A mouse surgical model for metastatic ovarian granulosa cell tumor.Comp Med. 2009 Dec;59(6):553-6. Comp Med. 2009. PMID: 20034430 Free PMC article.
-
Chronic restraint stress induces excessive activation of primordial follicles in mice ovaries.PLoS One. 2018 Mar 30;13(3):e0194894. doi: 10.1371/journal.pone.0194894. eCollection 2018. PLoS One. 2018. PMID: 29601583 Free PMC article.
References
-
- Jubb KVF, et al. Pathology of Domestic Animals. 4th edn. San Diego, CA: American Press; 1993.
-
- Schumer ST, et al. Granulosa cell tumor of the ovary. J. Clin. Oncol. 2003;21:1180–1189. - PubMed
-
- Villella J, et al. Clinical and pathological predictive factors in women with adult-type granulosa cell tumor of the ovary. Int. J. Gynecol. Pathol. 2007;26:154–159. - PubMed
-
- East N, et al. Granulosa cell tumour: a recurrence 40 years after initial diagnosis. J. Obstet. Gynaecol. Can. 2005;27:363–364. - PubMed
-
- Boerboom D, et al. Misregulated Wnt/beta-catenin signaling leads to ovarian granulosa cell tumor development. Cancer Res. 2005;65:9206–9215. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

