Vascular endothelial growth factor mediates the estrogen-induced breakdown of tight junctions between and increase in proliferation of microvessel endothelial cells in the baboon endometrium
- PMID: 18687775
- PMCID: PMC2613057
- DOI: 10.1210/en.2008-0521
Vascular endothelial growth factor mediates the estrogen-induced breakdown of tight junctions between and increase in proliferation of microvessel endothelial cells in the baboon endometrium
Abstract
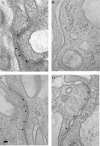
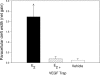
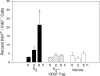
To assess whether there is a link between estrogen, vascular endothelial growth factor (VEGF), and early aspects of uterine angiogenesis, an acute temporal study was conducted in which ovariectomized baboons were pretreated with VEGF Trap, which sequesters endogenous VEGF, and administered estradiol at time 0 h. Serum estradiol levels approximated 500 pg/ml 4-6 h after estradiol administration. VEGF mRNA levels in endometrial glandular epithelial and stromal cells were increased to values 6 h after estradiol that were 3.74 +/- 0.99-fold (mean +/- se) and 5.70 +/- 1.60-fold greater (P < 0.05), respectively, than at 0 h. Microvessel interendothelial cell tight junctions, which control paracellular permeability, were present in the endometrium at time 0 h, but not evident 6 h after estradiol administration. Thus, microvessel paracellular cleft width increased (P < 0.01, ANOVA) from 5.03 +/- 0.22 nm at 0 h to 7.27 +/- 0.48 nm 6 h after estrogen. In contrast, tight junctions remained intact, and paracellular cleft widths were unaltered in estradiol/VEGF Trap and vehicle-treated animals. Endometrial microvessel endothelial cell mitosis, i.e. percent Ki67+/Ki67- immunolabeled endothelial cells, increased (P < 0.05) from 2.9 +/- 0.3% at 0 h to 21.4 +/- 7.0% 6 h after estrogen treatment but was unchanged in estradiol/VEGF Trap and vehicle-treated animals. In summary, the estrogen-induced disruption of endometrial microvessel endothelial tight junctions and increase in endothelial cell proliferation were prevented by VEGF Trap. Therefore, we propose that VEGF mediates the estrogen-induced increase in microvessel permeability and endothelial cell proliferation as early steps in angiogenesis in the primate endometrium.
Figures



References
-
- Brenner RM, Slayden OD 1994 Cyclic changes in the primate oviduct and endometrium. In: Knobil E, Neill JD, eds. The physiology of reproduction. 2nd ed. New York: Raven Press; 541–569
-
- Albrecht ED, Pepe GJ 2003 Steroid hormone regulation of angiogenesis in the primate endometrium: progesterone action and the endometrium. Front Biosci 8:d416–d429 - PubMed
-
- Girling JE, Rogers PAW 2005 Recent advances in endometrial angiogenesis research. Angiogenesis 8:89–99 - PubMed
-
- Dvorak HF, Nagy JA, Feng D, Brown LF, Dvorak AM 1999 Vascular permeability factor/vascular endothelial growth factor and the significance of microvascular hyperpermeability in angiogenesis. Curr Top Microbiol Immunol 237:97–132 - PubMed
-
- Dejana E, Spagnuolo R, Bazzoni G 2001 Interendothelial junctions and their role in the control of angiogenesis, vascular permeability and leukocyte transmigration. Thromb Haemost 86:308–315 - PubMed

