CCR2 receptor is essential to activate microbicidal mechanisms to control Toxoplasma gondii infection in the central nervous system
- PMID: 18688032
- PMCID: PMC2527091
- DOI: 10.2353/ajpath.2008.080129
CCR2 receptor is essential to activate microbicidal mechanisms to control Toxoplasma gondii infection in the central nervous system
Abstract
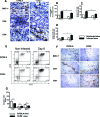
Chemokines comprise a structurally related family of cytokines that regulate leukocyte trafficking. Because infection with Toxoplasma gondii can induce an important inflammatory reaction that, if left uncontrolled, can lead to death, we investigated the role of the chemokine receptor CCR2 in T. gondii infection. We orally infected CCR2(-/-) mice with five ME-49 T. gondii cysts and monitored morbidity, survival, and immune response thereafter. The CCR2(-/-) mice displayed higher susceptibility to infection as all mice died on day 28 after infection. Despite similar Th1 responses, a more evident anti-inflammatory response was induced in the peripheral organs of CCR2(-/-) mice compared with wild-type C57BL/6 mice. Additionally, CCR2(-/-) mice presented greater parasitism and a milder inflammatory reaction in their peripheral organs with lesser CD4(+) and MAC-1(+) and greater CD8(+) cell migration. The parasite load decreased in these organs in CCR2(-/-) mice but remained uncontrolled in the central nervous system. Additionally, we observed down-regulated inducible nitric oxide synthase expression in peripheral organs from CCR2(-/-) mice that was associated with a small nitric oxide production by spleen macrophages. In conclusion, in the absence of CCR2, another mechanism is activated to control tissue parasitism in peripheral organs. Nevertheless, CCR2 is essential for the activation of microbicidal mediators that control T. gondii replication in the central nervous system.
Figures



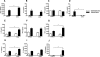

Similar articles
-
Insights into inflammatory bowel disease using Toxoplasma gondii as an infectious trigger.Immunol Cell Biol. 2012 Aug;90(7):668-75. doi: 10.1038/icb.2011.93. Epub 2011 Nov 8. Immunol Cell Biol. 2012. PMID: 22064707 Free PMC article. Review.
-
[Rudolf-Virchow Prize 1998. Award lecture. Toxoplasmosis: a model infection for studying systemic and intracerebral immune reactions].Verh Dtsch Ges Pathol. 1998;82:9-22. Verh Dtsch Ges Pathol. 1998. PMID: 10095413 German.
-
Interferon-gamma receptor-deficiency renders mice highly susceptible to toxoplasmosis by decreased macrophage activation.Lab Invest. 1996 Dec;75(6):827-41. Lab Invest. 1996. PMID: 8973478
-
Kinetics of systemic cytokine and brain chemokine gene expression in murine toxoplasma infection.J Parasitol. 2008 Dec;94(6):1282-8. doi: 10.1645/GE-1309.1. J Parasitol. 2008. PMID: 19127964
-
Small intestinal inflammation following oral infection with Toxoplasma gondii does not occur exclusively in C57BL/6 mice: review of 70 reports from the literature.Mem Inst Oswaldo Cruz. 2009 Mar;104(2):221-33. doi: 10.1590/s0074-02762009000200015. Mem Inst Oswaldo Cruz. 2009. PMID: 19430647 Review.
Cited by
-
NFAT1 Regulates Ly6Chi Monocyte Recruitment to the CNS and Plays an Essential Role in Resistance to Toxoplasma gondii Infection.Front Immunol. 2019 Sep 6;10:2105. doi: 10.3389/fimmu.2019.02105. eCollection 2019. Front Immunol. 2019. PMID: 31555297 Free PMC article.
-
Morphological and biochemical repercussions of Toxoplasma gondii infection in a 3D human brain neurospheres model.Brain Behav Immun Health. 2020 Dec 8;11:100190. doi: 10.1016/j.bbih.2020.100190. eCollection 2021 Feb. Brain Behav Immun Health. 2020. PMID: 34589727 Free PMC article.
-
Toxoplasma gondii infection and its implications within the central nervous system.Nat Rev Microbiol. 2021 Jul;19(7):467-480. doi: 10.1038/s41579-021-00518-7. Epub 2021 Feb 24. Nat Rev Microbiol. 2021. PMID: 33627834 Review.
-
Insights into inflammatory bowel disease using Toxoplasma gondii as an infectious trigger.Immunol Cell Biol. 2012 Aug;90(7):668-75. doi: 10.1038/icb.2011.93. Epub 2011 Nov 8. Immunol Cell Biol. 2012. PMID: 22064707 Free PMC article. Review.
-
Mouse adenovirus type 1 infection of macrophages.Virology. 2009 Aug 1;390(2):307-14. doi: 10.1016/j.virol.2009.05.025. Epub 2009 Jun 21. Virology. 2009. PMID: 19540545 Free PMC article.
References
-
- Brenier-Pinchart M-P, Villena I, Mercier C, Durand F, Simon J, Cesbron-Delauw M-F, Pelloux H. The Toxoplasma surface protein SAG1 triggers efficient in vitro secretion of chemokine ligand 2 (CCL2) from human fibroblasts. Microbes Infect. 2006;8:254–261. - PubMed
-
- Kasper L, Courret N, Darche S, Luangsay S, Mennechet LM, Rachinel N, Ronet C, Buzoni-Gatel D. Toxoplasma gondii and mucosal immunity. Int J Parasitol. 2004;34:401–409. - PubMed
-
- Rachinel N, Buzoni-Gatel D, Dutta C, Mennechet FJD, Luangsay S, Minns LA, Grigg ME, Tomavo S, Boothroyd JC, Kasper LH. The induction of acute ileitis by a single microbial antigen of Toxoplasma gondii. J Immunol. 2004;173:2725–2735. - PubMed
-
- Strack A, Asensio VC, Campbell IL, Schluter D, Deckert M. Chemokines are differentially expressed by astrocytes, microglia and inflammatory leukocytes in Toxoplasma encephalitis and critically regulated by interferon-gamma. Acta Neuropathol. 2002;103:458–468. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

