Pharmacoepidemiology of QT-interval prolonging drug administration in critically ill patients
- PMID: 18693297
- PMCID: PMC2587152
- DOI: 10.1002/pds.1637
Pharmacoepidemiology of QT-interval prolonging drug administration in critically ill patients
Abstract
Purpose: Commonly prescribed medications produce QT-prolongation and are associated with torsades de pointes in non-acutely ill patients. We examined patterns of QT-prolonging drug use in critically ill individuals.
Methods: An administrative critical care database was utilized to identify patients receiving drugs associated with QT-interval prolongation or torsades de pointes for > or = 24 hours.
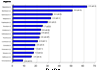
Results: Data from 212 016 individuals collected over a 63-month period was examined to identify 6125 patients (2.9%) receiving QT-interval prolonging drugs. These individuals had a mean (+/-SE) age of 63.0 (+/-0.2) years, were predominately male (55.4%) and Caucasian (84.4%), and were exposed to QT-interval prolonging agents for a mean (+/-SE) 53.1 (+/-0.4)% of their ICU length of stay. Respiratory and cardiovascular illnesses were the most common reasons for ICU admission (17.2, 12.0%, respectively). The most frequently administered agents were amiodarone (23.5%), haloperidol (19.8%), and levofloxacin (19.7%); no other single agent accounted for more than 10% of QT-interval prolonging drugs prescribed. Coadministration of QT-prolonging drugs occurred in 1139 patients (18.6%). These patients had higher ICU mortality rate and longer ICU lengths of stay, compared to patients not receiving coadministered drugs (p < 0.001 for both). For patients receiving coadministered drugs, overlap occurred for 71.4 (+/-0.8)% of the time that the drugs were given. Amiodarone coadministration with antibiotics, haloperidol coadministration with antibiotics, and haloperidol coadministration with amiodarone, comprised 15.2, 13.7, and 9.4%, of all coadministered agents, respectively.
Conclusions: QT-prolonging drugs were used in a minority of critically ill patients. Prospective evaluation in the ICU environment is necessary to determine whether administration of these agents is associated with adverse cardiac events comparable to those reported in ambulatory patients.
Figures

References
-
- Roden DM. Drug-induced prolongation of the QT interval. New England Journal of Medicine. 2004;350:1013–1022. - PubMed
-
- Curtis LH, Ostbye T, Sendersky V, Hutchison S, LaPointe NMA, Al-Khatib SM, et al. Prescription of QT-prolonging drugs in a cohort of about 5 million outpatients. American Journal of Medicine. 2003;114:135–141. - PubMed
-
- Al-Khatib SM, LaPointe NMA, Kramer JM, Califf RM. What clinicians should know about the QT interval. Journal of the American Medical Association. 2003;289:2120–2127. - PubMed
-
- Arizona Center for Education and Research on Therapeutics. [accessed 16 April 2008]. www.arizonacert.org.
-
- Smalley W, Shatin D, Wysowski DK, Gurwitz J, Andrade SE, Goodman M, et al. Contraindicated use of cisapride. Impact of Food and Drug Administration Regulatory Action. Journal of the American Medical Association. 2000;284:3036–3039. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

