Targeted microbubbles for imaging tumor angiogenesis: assessment of whole-body biodistribution with dynamic micro-PET in mice
- PMID: 18695212
- PMCID: PMC2657857
- DOI: 10.1148/radiol.2491072050
Targeted microbubbles for imaging tumor angiogenesis: assessment of whole-body biodistribution with dynamic micro-PET in mice
Abstract
Purpose: To evaluate in vivo whole-body biodistribution of microbubbles (MBs) targeted to tumor angiogenesis-related vascular endothelial growth factor (VEGF) receptor 2 (VEGFR2) by using dynamic micro-positron emission tomography (PET) in living mice.
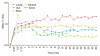
Materials and methods: Animal protocols were approved by the Institutional Administrative Panel on Laboratory Animal Care. Lipid-shell perfluorocarbon-filled MBs, targeted to VEGFR2 via anti-VEGFR2 antibodies, were radiolabeled by conjugating the radiofluorination agent N-succinimidyl-4-[(18)F]fluorobenzoate (SFB) to the anti-VEGFR2 antibodies. These MBs were then injected intravenously into nude mice (n = 4) bearing angiosarcomas, and the whole-body biodistribution of these probes was assessed for 60 minutes by using dynamic micro-PET. Results were compared with ex vivo gamma counting (n = 6) and immunofluorescence staining (n = 6). Control studies in angiosarcoma-bearing mice were performed with injection of the radiolabeled antibodies alone (n = 3) or free SFB (n = 3). A mixed-effects regression of MB accumulation on fixed effects of time and tissue type (tumor or muscle) and random effect of animal was performed.
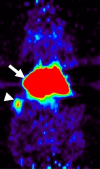
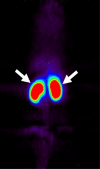
Results: VEGFR2-targeted MBs rapidly cleared from the blood circulation (50% blood clearance after approximately 3.5 minutes) and accumulated in the liver (mean, 33.4% injected dose [ID]/g +/- 13.7 [standard deviation] at 60 minutes) and spleen (mean, 9.3% ID/g +/- 6.5 at 60 minutes) on the basis of micro-PET imaging. These findings were confirmed with ex vivo gamma counting. Uptake of targeted MBs was significantly higher (P < .0001) in tumor than in adjacent skeletal muscle tissue. Immunofluorescence staining demonstrated accumulation of the targeted MBs within hepatic Kupffer cells and splenic macrophages. Biodistribution of the radiolabeled antibodies and free SFB differed from the distribution of the targeted MBs.
Conclusion: Dynamic micro-PET allows assessment of in vivo biodistribution of VEGFR2-targeted MBs.
(c) RSNA, 2008.
Figures


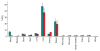
Similar articles
-
Dual-targeted contrast agent for US assessment of tumor angiogenesis in vivo.Radiology. 2008 Sep;248(3):936-44. doi: 10.1148/radiol.2483072231. Radiology. 2008. PMID: 18710985 Free PMC article.
-
US imaging of tumor angiogenesis with microbubbles targeted to vascular endothelial growth factor receptor type 2 in mice.Radiology. 2008 Feb;246(2):508-18. doi: 10.1148/radiol.2462070536. Epub 2008 Jan 7. Radiology. 2008. PMID: 18180339 Free PMC article.
-
Evaluation of 18F-labeled targeted perfluorocarbon-filled albumin microbubbles as a probe for microUS and microPET in tumor-bearing mice.Ultrasonics. 2013 Feb;53(2):320-7. doi: 10.1016/j.ultras.2012.06.014. Epub 2012 Jul 7. Ultrasonics. 2013. PMID: 22832082
-
18F-Labeled N-succinimidyl-4-fluorobenzoate–conjugated rat anti-mouse vascular endothelial growth factor receptor 2 monoclonal antibody linked to microbubbles.2011 Nov 14 [updated 2012 Mar 15]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. 2011 Nov 14 [updated 2012 Mar 15]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. PMID: 22439163 Free Books & Documents. Review.
-
Buried cyclo(Arg-Gly-Tyr(Me)-Lys-Glu) microbubbles.2008 Jun 2 [updated 2008 Jul 21]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. 2008 Jun 2 [updated 2008 Jul 21]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. PMID: 20641944 Free Books & Documents. Review.
Cited by
-
Quantitative Pharmacokinetics Reveal Impact of Lipid Composition on Microbubble and Nanoprogeny Shell Fate.Adv Sci (Weinh). 2024 Jan;11(4):e2304453. doi: 10.1002/advs.202304453. Epub 2023 Nov 30. Adv Sci (Weinh). 2024. PMID: 38032129 Free PMC article.
-
Aminolysis-mediated single-step surface functionalization of poly (butyl cyanoacrylate) microbubbles for ultrasound molecular imaging.J Nanobiotechnology. 2024 Sep 1;22(1):528. doi: 10.1186/s12951-024-02806-9. J Nanobiotechnology. 2024. PMID: 39218888 Free PMC article.
-
Molecular and clinical aspects of targeting the VEGF pathway in tumors.J Oncol. 2010;2010:652320. doi: 10.1155/2010/652320. Epub 2010 Jun 10. J Oncol. 2010. PMID: 20628530 Free PMC article.
-
Validation of Normalized Singular Spectrum Area as a Classifier for Molecularly Targeted Microbubble Adherence.Ultrasound Med Biol. 2019 Sep;45(9):2493-2501. doi: 10.1016/j.ultrasmedbio.2019.05.026. Epub 2019 Jun 19. Ultrasound Med Biol. 2019. PMID: 31227262 Free PMC article.
-
Assessment and monitoring tumor vascularity with contrast-enhanced ultrasound maximum intensity persistence imaging.Invest Radiol. 2011 Mar;46(3):187-95. doi: 10.1097/RLI.0b013e3181f9202d. Invest Radiol. 2011. PMID: 21150790 Free PMC article.
References
-
- Lindner JR. Microbubbles in medical imaging: current applications and future directions. Nat Rev Drug Discov 2004;3:527–532. - PubMed
-
- Willmann JK, van Bruggen N, Dinkelborg LM, Gambhir SS. Molecular imaging in drug development. Nat Rev Drug Discov 2008;7:591–607. - PubMed
-
- Ellegala DB, Leong-Poi H, Carpenter JE, et al. Imaging tumor angiogenesis with contrast ultrasound and microbubbles targeted to alpha(v)beta3. Circulation 2003;108:336–341. - PubMed
-
- Korpanty G, Carbon JG, Grayburn PA, Fleming JB, Brekken RA. Monitoring response to anticancer therapy by targeting microbubbles to tumor vasculature. Clin Cancer Res 2007;13:323–330. - PubMed
-
- Weller GE, Wong MK, Modzelewski RA, et al. Ultrasonic imaging of tumor angiogenesis using contrast microbubbles targeted via the tumor-binding peptide arginine-arginine-leucine. Cancer Res 2005;65:533–539. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

