Cross-talk between G protein-coupled and epidermal growth factor receptors regulates gonadotropin-mediated steroidogenesis in Leydig cells
- PMID: 18701461
- PMCID: PMC2562062
- DOI: 10.1074/jbc.M803867200
Cross-talk between G protein-coupled and epidermal growth factor receptors regulates gonadotropin-mediated steroidogenesis in Leydig cells
Abstract
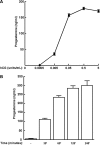
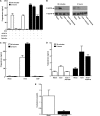
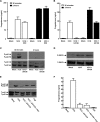
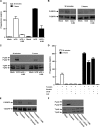
Gonadal steroid production is stimulated by gonadotropin binding to G protein-coupled receptors (GPCRs). Although GPCR-mediated increases in intracellular cAMP are known regulators of steroidogenesis, the roles of other signaling pathways in mediating steroid production are not well characterized. Recent studies suggest that luteinizing hormone (LH) receptor activation leads to trans-activation of epidermal growth factor (EGF) receptors in the testes and ovary. This pathway is critical for LH-induced steroid production in ovarian follicles, probably through matrix metalloproteinase (MMP)-mediated release of EGF receptor (EGFR) binding ectodomains. Here we examined LH and EGF receptor cross-talk in testicular steroidogenesis using mouse MLTC-1 Leydig cells. We demonstrated that, similar to the ovary, trans-activation of the EGF receptor was critical for gonadotropin-induced steroid production in Leydig cells. LH-induced increases in cAMP and cAMP-dependent protein kinase (PKA) activity mediated trans-activation of the EGF receptor and subsequent mitogen-activated protein kinase (MAPK) activation, ultimately leading to StAR phosphorylation and mitochondrial translocation. Steroidogenesis in Leydig cells was unaffected by MMP inhibitors, suggesting that cAMP and PKA trans-activated EGF receptors in an intracellular fashion. Interestingly, although cAMP was always needed for steroidogenesis, the EGFR/MAPK pathway was activated and necessary only for early (30-60 min), but not late (120 min or more), LH-induced steroidogenesis in vitro. In contrast, 36-h EGF receptor inhibition in vivo significantly reduced serum testosterone levels in male mice, demonstrating the physiologic importance of this cross-talk. These results suggest that GPCR-EGF receptor cross-talk is a conserved regulator of gonadotropin-induced steroidogenesis in the gonads, although the mechanisms of EGF receptor trans-activation may vary.
Figures

Similar articles
-
GPCR/EGFR cross talk is conserved in gonadal and adrenal steroidogenesis but is uniquely regulated by matrix metalloproteinases 2 and 9 in the ovary.Mol Endocrinol. 2011 Jun;25(6):1055-65. doi: 10.1210/me.2010-0410. Epub 2011 Mar 31. Mol Endocrinol. 2011. PMID: 21454403 Free PMC article.
-
cAMP-independent signaling regulates steroidogenesis in mouse Leydig cells in the absence of StAR phosphorylation.J Mol Endocrinol. 2006 Aug;37(1):81-95. doi: 10.1677/jme.1.02065. J Mol Endocrinol. 2006. PMID: 16901926
-
Membrane receptor cross talk in gonadotropin-, IGF-I-, and insulin-mediated steroidogenesis in fish ovary: An overview.Gen Comp Endocrinol. 2017 Jan 1;240:10-18. doi: 10.1016/j.ygcen.2016.09.002. Epub 2016 Sep 8. Gen Comp Endocrinol. 2017. PMID: 27616426
-
Membrane receptor cross talk in steroidogenesis: recent insights and clinical implications.Steroids. 2013 Jun;78(6):633-8. doi: 10.1016/j.steroids.2012.12.016. Epub 2013 Feb 1. Steroids. 2013. PMID: 23380369 Review.
-
Hormonal regulation of androgen production by the Leydig cell.J Steroid Biochem. 1984 Jan;20(1):161-73. doi: 10.1016/0022-4731(84)90203-6. J Steroid Biochem. 1984. PMID: 6323862 Review.
Cited by
-
LH-Induced Steroidogenesis in the Mouse Ovary, but Not Testis, Requires Matrix Metalloproteinase 2- and 9-Mediated Cleavage of Upregulated EGF Receptor Ligands.Biol Reprod. 2015 Sep;93(3):65. doi: 10.1095/biolreprod.115.130971. Epub 2015 Jul 22. Biol Reprod. 2015. PMID: 26203177 Free PMC article.
-
Epidermal growth factor regulates the development of stem and progenitor Leydig cells in rats.J Cell Mol Med. 2020 Jul;24(13):7313-7330. doi: 10.1111/jcmm.15302. Epub 2020 May 22. J Cell Mol Med. 2020. PMID: 32441057 Free PMC article.
-
Luteinizing hormone/human chorionic gonadotropin-mediated activation of mTORC1 signaling is required for androgen synthesis by theca-interstitial cells.Mol Endocrinol. 2012 Oct;26(10):1732-42. doi: 10.1210/me.2012-1106. Epub 2012 Jul 24. Mol Endocrinol. 2012. PMID: 22827930 Free PMC article.
-
Gonadotoxicity of immunotherapy and targeted agents in patients with cancer and impact on subsequent pregnancies.Hum Reprod. 2025 Aug 1;40(8):1452-1466. doi: 10.1093/humrep/deaf096. Hum Reprod. 2025. PMID: 40482082 Free PMC article. Review.
-
Paxillin regulates androgen- and epidermal growth factor-induced MAPK signaling and cell proliferation in prostate cancer cells.J Biol Chem. 2010 Sep 10;285(37):28787-95. doi: 10.1074/jbc.M110.134064. Epub 2010 Jul 13. J Biol Chem. 2010. PMID: 20628053 Free PMC article.
References
-
- Scaglia, H. E., Carrere, C. A., Mariani, V. A., Zylbersztein, C. C., Rey-Valzacchi, G. J., Kelly, E. E., and Aquilano, D. R. (1991) J. Androl. 12 273–280 - PubMed
-
- Talbot, J. A., Rodger, R. S., Shalet, S. M., Littley, M. D., and Robertson, W. R. (1990) Acta Endocrinol. (Copenh.) 122 643–650 - PubMed
-
- Podesta, E. J., Dufau, M. L., and Catt, K. J. (1978) Biochemistry 17 1566–1573 - PubMed
-
- Stocco, D. M., and Chaudhary, L. R. (1990) Cell. Signal. 2 161–170 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

