Suppression of inhibitor of differentiation 2, a target of mutant p53, is required for gain-of-function mutations
- PMID: 18701504
- PMCID: PMC2597213
- DOI: 10.1158/0008-5472.CAN-08-0810
Suppression of inhibitor of differentiation 2, a target of mutant p53, is required for gain-of-function mutations
Abstract
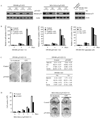
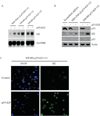
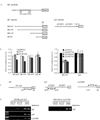
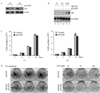
Overexpression of mutant p53 is a common theme in human tumors, suggesting a tumor-promoting gain-of-function for mutant p53. To elucidate whether and how mutant p53 acquires its gain-of-function, mutant p53 is inducibly knocked down in the SW480 colon cancer cell line, which contains mutant p53(R273H/P309S), and the MIA PaCa-2 pancreatic cancer cell line, which contains mutant p53(R248W). We found that knockdown of mutant p53 markedly inhibits cell proliferation. In addition, knockdown of mutant p53 sensitizes tumor cells to growth suppression by various chemotherapeutic drugs. To determine whether a gene involved in cell growth and survival is regulated by mutant p53, gene expression profiling analysis was performed and showed that the expression level of Id2, a member of the inhibitor of differentiation (Id) family, was markedly increased upon knockdown of mutant p53. To confirm this, Northern blot analysis was performed and showed that the expression level of Id2 was regulated by various mutant p53s in multiple cell lines. In addition, we found that the Id2 promoter is responsive to mutant but not wild-type p53, and mutant p53 binds to the Id2 promoter. Consistent with these observations, expression of endogenous Id2 was found to be inhibited by exogenous mutant p53 in p53-null HCT116 cells. Finally, we showed that knockdown of Id2 can restore the proliferative potential of tumor cells inhibited by withdrawal of mutant p53. Together, these findings suggest that one mechanism by which mutant p53 acquires its gain-of-function is through the inhibition of Id2 expression.
Figures

Similar articles
-
Characterization of functional domains necessary for mutant p53 gain of function.J Biol Chem. 2010 May 7;285(19):14229-38. doi: 10.1074/jbc.M109.097253. Epub 2010 Mar 8. J Biol Chem. 2010. PMID: 20212049 Free PMC article.
-
Identification of GRO1 as a critical determinant for mutant p53 gain of function.J Biol Chem. 2009 May 1;284(18):12178-87. doi: 10.1074/jbc.M900994200. Epub 2009 Mar 3. J Biol Chem. 2009. PMID: 19258312 Free PMC article.
-
Mutant p53-induced up-regulation of mitogen-activated protein kinase kinase 3 contributes to gain of function.J Biol Chem. 2010 May 7;285(19):14160-9. doi: 10.1074/jbc.M109.094813. Epub 2010 Mar 11. J Biol Chem. 2010. PMID: 20223820 Free PMC article.
-
p53 regulates FAK expression in human tumor cells.Mol Carcinog. 2008 May;47(5):373-82. doi: 10.1002/mc.20395. Mol Carcinog. 2008. PMID: 17999388 Free PMC article.
-
Repression of Id2 expression by Gfi-1 is required for B-cell and myeloid development.Blood. 2010 Aug 19;116(7):1060-9. doi: 10.1182/blood-2009-11-255075. Epub 2010 May 7. Blood. 2010. PMID: 20453161 Free PMC article.
Cited by
-
Effects of a histone deacetylase inhibitor, sodium butyrate, on 53-kDa protein expression and sensitivity to anticancer drugs of pancreatic cancer cells.Curr Ther Res Clin Exp. 2010 Jun;71(3):162-72. doi: 10.1016/j.curtheres.2010.06.002. Curr Ther Res Clin Exp. 2010. PMID: 24683262 Free PMC article.
-
Understanding Disease Biology and Informing the Management of Pancreas Cancer With Preclinical Model Systems.Cancer J. 2017 Nov/Dec;23(6):326-332. doi: 10.1097/PPO.0000000000000289. Cancer J. 2017. PMID: 29189328 Free PMC article. Review.
-
An image-based, high-throughput screening assay for molecules that induce excess DNA replication in human cancer cells.Mol Cancer Res. 2011 Mar;9(3):294-310. doi: 10.1158/1541-7786.MCR-10-0570. Epub 2011 Jan 21. Mol Cancer Res. 2011. PMID: 21257818 Free PMC article.
-
MiR-135 suppresses glycolysis and promotes pancreatic cancer cell adaptation to metabolic stress by targeting phosphofructokinase-1.Nat Commun. 2019 Feb 18;10(1):809. doi: 10.1038/s41467-019-08759-0. Nat Commun. 2019. PMID: 30778058 Free PMC article.
-
p53 directly represses Id2 to inhibit the proliferation of neural progenitor cells.Stem Cells. 2011 Jul;29(7):1090-101. doi: 10.1002/stem.660. Stem Cells. 2011. PMID: 21608079 Free PMC article.
References
-
- Loehberg CR, Thompson T, Kastan MB, et al. Ataxia telangiectasia-mutated and p53 are potential mediators of chloroquine-induced resistance to mammary carcinogenesis. Cancer Res. 2007;67:12026–12033. - PubMed
-
- Lang GA, Iwakuma T, Suh YA, et al. Gain of function of a p53 hot spot mutation in a mouse model of Li-Fraumeni syndrome. Cell. 2004;119:861–872. - PubMed
-
- Bossi G, Lapi E, Strano S, Rinaldo C, Blandino G, Sacchi A. Mutant p53 gain of function: reduction of tumor malignancy of human cancer cell lines through abrogation of mutant p53 expression. Oncogene. 2006;25:304–309. - PubMed
-
- Olive KP, Tuveson DA, Ruhe ZC, et al. Mutant p53 gain of function in two mouse models of Li-Fraumeni syndrome. Cell. 2004;119:847–860. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

