Critical role of PPAR-alpha in perfluorooctanoic acid- and perfluorodecanoic acid-induced downregulation of Oatp uptake transporters in mouse livers
- PMID: 18703564
- PMCID: PMC2563139
- DOI: 10.1093/toxsci/kfn161
Critical role of PPAR-alpha in perfluorooctanoic acid- and perfluorodecanoic acid-induced downregulation of Oatp uptake transporters in mouse livers
Abstract
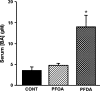
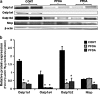
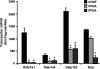
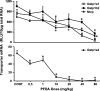
Perfluorooctanoic acid (PFOA) and perfluorodecanoic acid (PFDA) have been detected globally in wildlife and humans. Data from a gene array indicate that PFOA decreases organic anion transporting polypeptides (Oatps) in liver. Na(+)-taurocholate cotransporting polypeptide (Ntcp) and Oatp1a1, 1a4, and 1b2 are major transporters responsible for uptake of bile acids (BAs) and other organic compounds into liver. The purpose of the present study was to determine the effects of two perfluorinated fatty acids, PFOA and PFDA, on mRNA and protein expression of hepatic uptake transporters Oatps and Ntcp, and to determine the underlying regulatory mechanisms by using peroxisome proliferator-activated receptor alpha (PPAR-alpha), constitutive androstane receptor, pregnane-X receptor, NF-E2-related factor 2, and farnesoid X receptor-null mouse models. After 2 days following a single i.p. administration, PFOA did not alter serum BA concentrations, but PFDA increased serum BA concentrations 300%. Furthermore, PFOA decreased mRNA and protein expression of Oatp1a1, 1a4, and 1b2, but not Ntcp in mouse liver. In contrast, PFDA decreased mRNA and protein expression of all four transporters, and decreased the mRNA expression in a dose-dependent manner, with the decrease of Oatp1a4 occurring at lower doses than the other three transporters. Multiple mechanisms are likely involved in the down-regulation of mouse Oatps and Ntcp by PFDA. By using the various transcription factor-null mice, PPAR-alpha was shown to play a central role in the down-regulation of Oatp1a1, 1a4, 1b2, and Ntcp by PFDA. The current studies provide important insight into understanding the mechanisms by which PFDA regulate the expression of hepatic uptake transporters. In conclusion, PFOA and PFDA decrease mouse liver uptake transporters primarily via activation of PPAR-alpha.
Figures
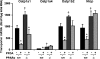
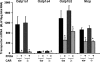
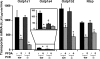
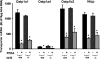
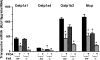
References
-
- Abdellatif AG, Preat V, Taper HS, Roberfroid M. The modulation of rat liver carcinogenesis by perfluorooctanoic acid, a peroxisome proliferator. Toxicol. Appl. Pharmacol. 1991;111:530–537. - PubMed
-
- Adinehzadeh M, Reo NV. Effects of peroxisome proliferators on rat liver phospholipids: Sphingomyelin degradation may be involved in hepatotoxic mechanism of perfluorodecanoic acid. Chem. Res. Toxicol. 1998;11:428–440. - PubMed
-
- Akiyama TE, Nicol CJ, Fievet C, Staels B, Ward JM, Auwerx J, Lee SS, Gonzalez FJ, Peters JM. Peroxisome proliferator-activated receptor-alpha regulates lipid homeostasis, but is not associated with obesity: Studies with congenic mouse lines. J. Biol. Chem. 2001;276:39088–39093. - PubMed
-
- Aleksunes LM, Scheffer GL, Jakowski AB, Pruimboom-Brees IM, Manautou JE. Coordinated expression of multidrug resistance-associated proteins (Mrps) in mouse liver during toxicant-induced injury. Toxicol. Sci. 2006;89:370–379. - PubMed
-
- Bookstaff RC, Moore RW, Ingall GB, Peterson RE. Androgenic deficiency in male rats treated with perfluorodecanoic acid. Toxicol. Appl. Pharmacol. 1990;104:322–333. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

