GAPDH is conformationally and functionally altered in association with oxidative stress in mouse models of amyotrophic lateral sclerosis
- PMID: 18706911
- PMCID: PMC6348903
- DOI: 10.1016/j.jmb.2008.07.088
GAPDH is conformationally and functionally altered in association with oxidative stress in mouse models of amyotrophic lateral sclerosis
Abstract
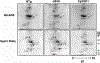
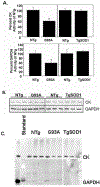
It is proposed that conformational changes induced in proteins by oxidation can lead to loss of activity or protein aggregation through exposure of hydrophobic residues and alteration in surface hydrophobicity. Because increased oxidative stress and protein aggregation are consistently observed in amyotrophic lateral sclerosis (ALS), we used a 4,4'-dianilino-1,1'-binaphthyl-5,5'-disulfonic acid (BisANS) photolabeling approach to monitor changes in protein unfolding in vivo in skeletal muscle proteins in ALS mice. We find two major proteins, creatine kinase (CK) and glyceraldehyde-3-phosphate dehydrogenase (GAPDH), conformationally affected in the ALS G93A mouse model concordant with a 43% and 41% reduction in enzyme activity, respectively. This correlated with changes in conformation and activity that were detected in CK and GAPDH with in vitro oxidation. Interestingly, we found that GAPDH, but not CK, is conformationally and functionally affected in a longer-lived ALS model (H46R/H48Q), exhibiting a 22% reduction in enzyme activity. We proposed a reaction mechanism for BisANS with nucleophilic amino acids such as lysine, serine, threonine, and tyrosine, and BisANS was found to be primarily incorporated to lysine residues in GAPDH. We identified the specific BisANS incorporation sites on GAPDH in nontransgenic (NTg), G93A, and H46R/H48Q mice using liquid chromatography-tandem mass spectrometry analysis. Four BisANS-containing sites (K52, K104, K212, and K248) were found in NTg GAPDH, while three out of four of these sites were lost in either G93A or H46R/H48Q GAPDH. Conversely, eight new sites (K2, K63, K69, K114, K183, K251, S330, and K331) were found on GAPDH for G93A, including one common site (K114) for H46R/H48Q, which is not found on GAPDH from NTg mice. These data show that GAPDH is differentially affected structurally and functionally in vivo in accordance with the degree of oxidative stress associated with these two models of ALS.
Conflict of interest statement
Competing Financial Interests Statement: N/A
Figures


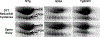





Similar articles
-
Heat shock factor 1 over-expression protects against exposure of hydrophobic residues on mutant SOD1 and early mortality in a mouse model of amyotrophic lateral sclerosis.Mol Neurodegener. 2013 Nov 21;8:43. doi: 10.1186/1750-1326-8-43. Mol Neurodegener. 2013. PMID: 24256636 Free PMC article.
-
A novel approach for screening the proteome for changes in protein conformation.Biochemistry. 2006 Mar 7;45(9):3077-85. doi: 10.1021/bi052031i. Biochemistry. 2006. PMID: 16503663
-
Differential effects of mutant SOD1 on protein structure of skeletal muscle and spinal cord of familial amyotrophic lateral sclerosis: role of chaperone network.Biochem Biophys Res Commun. 2013 Aug 16;438(1):218-23. doi: 10.1016/j.bbrc.2013.07.060. Epub 2013 Jul 23. Biochem Biophys Res Commun. 2013. PMID: 23886956
-
Oxidatively modified glyceraldehyde-3-phosphate dehydrogenase in neurodegenerative processes and the role of low molecular weight compounds in counteracting its aggregation and nuclear translocation.Ageing Res Rev. 2018 Dec;48:21-31. doi: 10.1016/j.arr.2018.09.003. Epub 2018 Sep 22. Ageing Res Rev. 2018. PMID: 30254002 Review.
-
[The biological significance of oxidative modifications of cysteine residues in proteins illustrated with the example of glyceraldehyde-3-phosphate dehydrogenase].Postepy Hig Med Dosw (Online). 2014 Mar 12;68:280-90. doi: 10.5604/17322693.1093929. Postepy Hig Med Dosw (Online). 2014. PMID: 24662796 Review. Polish.
Cited by
-
Reduced mitochondrial ROS, enhanced antioxidant defense, and distinct age-related changes in oxidative damage in muscles of long-lived Peromyscus leucopus.Am J Physiol Regul Integr Comp Physiol. 2013 Mar 1;304(5):R343-55. doi: 10.1152/ajpregu.00139.2012. Epub 2013 Jan 16. Am J Physiol Regul Integr Comp Physiol. 2013. PMID: 23325454 Free PMC article.
-
Derivation and validation of an epigenetic frailty risk score in population-based cohorts of older adults.Nat Commun. 2022 Sep 7;13(1):5269. doi: 10.1038/s41467-022-32893-x. Nat Commun. 2022. PMID: 36071044 Free PMC article.
-
The Nitrobenzoxadiazole Derivative NBDHEX Behaves as Plasmodium falciparum Gametocyte Selective Inhibitor with Malaria Parasite Transmission Blocking Activity.Pharmaceuticals (Basel). 2022 Jan 29;15(2):168. doi: 10.3390/ph15020168. Pharmaceuticals (Basel). 2022. PMID: 35215282 Free PMC article.
-
Antioxidant Effects and Mechanisms of Medicinal Plants and Their Bioactive Compounds for the Prevention and Treatment of Type 2 Diabetes: An Updated Review.Oxid Med Cell Longev. 2020 Feb 13;2020:1356893. doi: 10.1155/2020/1356893. eCollection 2020. Oxid Med Cell Longev. 2020. PMID: 32148647 Free PMC article. Review.
-
Glyceraldehyde-3-phosphate Dehydrogenase (GAPDH) Aggregation Causes Mitochondrial Dysfunction during Oxidative Stress-induced Cell Death.J Biol Chem. 2017 Mar 17;292(11):4727-4742. doi: 10.1074/jbc.M116.759084. Epub 2017 Feb 6. J Biol Chem. 2017. PMID: 28167533 Free PMC article.
References
-
- Widmer R, Ziaja I & Grune T (2006). Protein oxidation and degradation during aging: role in skin aging and neurodegeneration. Free Radic Res 40, 1259–68. - PubMed
-
- Stadtman ER (2006). Protein oxidation and aging. Free Radic Res 40, 1250–8. - PubMed
-
- Stadtman ER (2001). Protein oxidation in aging and age-related diseases. Ann N Y Acad Sci 928, 22–38. - PubMed
-
- Khidekel N & Hsieh-Wilson LC (2004). A ‘molecular switchboard’--covalent modifications to proteins and their impact on transcription. Org Biomol Chem 2, 1–7. - PubMed
-
- Nielsen ML, Savitski MM & Zubarev RA (2006). Extent of modifications in human proteome samples and their effect on dynamic range of analysis in shotgun proteomics. Mol Cell Proteomics 5, 2384–91. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

