A computational characterization of boron-oxygen multiple bonding in HN=CH-CH=CH-NH-BO
- PMID: 18707068
- PMCID: PMC2559978
- DOI: 10.1021/jp800125p
A computational characterization of boron-oxygen multiple bonding in HN=CH-CH=CH-NH-BO
Abstract
Structures, relative energies, and bonding characteristics for various conformers of 3-imino-N-(oxoboryl)prop-1-en-1-amine, HN=CH-CH=CH-NH-BO, and the corresponding borocycle (-HN=CH-CH=CH-NH-B-)O are discussed using results from second-order Møller-Plesset (MP2) perturbation theory with the Dunning-Woon correlation-consistent cc-pVDZ, aug-cc-pVDZ, and cc-pVTZ basis sets. These MP2 results are compared to those from computationally efficient density functional theory (DFT) calculations using the LDA, PBE, TPSS, BLYP, B3LYP, BVP86, OLYP, O3LYP, and PBE1PBE functionals in conjunction with the economical Pople-type 6-311++G(d,p) basis set to evaluate the suitability of these DFT/6-311++G(d,p) levels for use with larger boron-containing systems. The effects of an aqueous environment were incorporated into the calculations using COSMO methodology. The calculated boron-oxygen bond lengths, orbital compositions, and bond orders in all the (acyclic) HN=CH-CH=CH-NH-BO conformers were consistent with the presence of a boron-oxygen triple bond, similar to that found in H-BO and H2N-BO. The (-HN=CH-CH=CH-NH-B-)O borocycle is predicted to be planar (C2v symmetry), and it is approximately 30 kcal/mol lower in energy than any of the (acyclic) HN=CH-CH=CH-NH-BO conformers; the boron-oxygen bond in this borocycle has significant double bond character, a bonding scheme for which there has been only one experimental structure reported in the literature (Vidovic, D. ; et al. J. Am. Chem. Soc. 2005, 127, 4566- 4569).
Figures

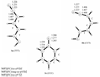
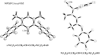

References
-
- Li Q, Xue F, Mak TCW. Crystalline inclusion compounds of urea with oxoboron components. Stabilization of the elusive dihydrogen borate anion in a hydrogen-bonded host lattice. Inorg. Chem. 1999;38:4142–4145.
-
- Ali HA, Dembitsy V, Srebnic M. Contemporary Aspects of Boron Chemistry and Biological Applications. Elsevier. 2005
-
- Hall DG. Boronic Acids: Preparation and Applications in Organic Synthesis and Medicine. John Wiley and Sons, Inc; 2005.
-
- Blacquiere JM, Sicora O, Vogels CM, Cuperlovic-culf M, Decken A, Ouellette RJ, Westcott SA. Dihydropyrimidinones containing boronic acids. Can. J. Chem. 2005;83:2052–2059.
-
- Malde AK, Khedkar SA, Coutino EC. The B(OH)−NH analog is a surrogate for the amide bond (CO-NH) in peptides: an ab initio study. J. Chem. Theory Comput. 2007;3:619–627. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

